Search
Search
Changing our MINDS
Local youth use art and storytelling to illustrate the personal and system challenges in mental health.
The words and images are both heartbreaking and hopeful.
“Nobody likes you if you are sad.”
you are enough. stay strong. let life surprise you.
“Please don’t leave like everyone else.”
1 year sober. 1 year of school. 1 year building a safe home. 1 year loving myself.
Zine-writing, a personal and introspective medium often used by people who have been marginalized, is an important part of local research that is finding solutions to complex mental health challenges among young adults. Its name derives from its magazine (“zine”) format.
“When we make the voices of people with lived experience central to our research, we can learn from each other and then change the system together,” says Dr. Arlene MacDougall, founder of MINDS of London-Middlesex.
The social innovation and research lab based at St. Joseph’s Health Care London (St. Joseph’s) is dedicated to designing, piloting and testing mental health innovations for teens and young adults. Its recent evolution into MINDS 2.0 adds insights by and for people up to age 35 who have complex, pervasive mental health and addiction issues.
Learning from experts
Rin, a London artist, who has published independent zines for a decade, created three for this project, including one that, with irony, asks the reader to ponder which vulnerable people aren’t worth saving.
“I believe making art can be very healing,” says Rin. “I wanted to share some of my story to help myself and help others. I want to play a part in shaping a mental health system that’s better than the one I encountered.”
The zines weave art, prose and poetry into story:
3 things I would change: affordability, stigma, waitlists.
“If I had a magic wand, I would use it to heal the waters, grow the trees, foster animal growth.”
Look in the mirror, see how far you’ve come.
They also raise and propose solutions to troubling issues among people with mental illness: housing and homelessness, sparse resources for people with addictions, lack of coordination among service providers, and dehumanizing attitudes in health and criminal justice systems.
“Participants experienced catharsis just in telling their stories,” says Renee Hunt PhD, Associate Director of Research and Operations at MINDS. “And because they’re the experts, they’re also playing an important part in changing systems that need an overhaul.”
Adds Rin, “It feels empowering knowing people are listening to us – people who care and are committed to making change to the system.”
MacDougall notes the pivotal role of St. Joseph’s Health Care Foundation – in particular, a groundbreaking $5-million donation towards mental health research from philanthropist Ryan Finch – in advancing mental health innovation through MINDS and MINDS 2.0.
“They’ve been our biggest supporters since day one. This wouldn’t have happened without support from Ryan and the foundation and all the many donors who believe in mental health research,” says MacDougall, who is also Director of Research and Innovation with St. Joseph’s Mental Health Care Program.
“MINDS has been a catalyst for system change,” MacDougall adds. “Research is always about finding answers and generating impact.”
Many findings from MINDS research have been put into practice, among them are the creation of guidebooks for peer support, free taxi service for rural youth needing urban mental health or addiction services, and educational resources to support 2SLGBTQI+ students.
MacDougall says MINDS 2.0 expands that work with new voices and the exploration of more in-depth solutions.
What’s next for MINDS 2.0
- Workshopping ideas and prototypes for mental health systems change. Sessions will be held where people with lived/living experience and service providers propose and develop potential solutions.
- Leadership-building. Twenty mental health practitioners will be trained to become “agents of change” who will collectively create, implement and evaluate mental health programs, technology, interventions and training.
- Imaginarium conference. In 2025, a first-of-its-kind national conference will take place to share system innovations in mental health and addiction.
Clinical trial will evaluate new therapy for patients with treatment-resistant depression as a result of bipolar disorder
LONDON, ON – Researchers at Lawson Health Research Institute are offering new hope to patients with treatment-resistant depression through participation in a national clinical trial. The study is the first randomized controlled trial to examine the efficacy of a new treatment called magnetic seizure therapy (MST) for patients with treatment-resistant depression as a result of bipolar disorder.
Treatment-resistant depression is a severe form of depression that does not respond to traditional therapies like medication. It is particularly common in patients with depression from bipolar disorder who are left with limited treatment options.
For years electroconvulsive therapy (ECT) has been the gold standard for patients with treatment-resistant depression. ECT uses an electric field to induce a seizure that provides a therapeutic benefit. But while ECT is effective, many patients opt out of treatment due to stigma surrounding the therapy and the potential for cognitive side effects like disorientation and amnesia.
MST has emerged as a promising alternative. MST works in a similar way to ECT but uses a focused magnetic field as opposed to electricity. As a result, MST induces a more focused seizure to reduce the risk of cognitive side effects.
“Magnetic seizure therapy has already been shown as a promising treatment for major depressive disorder or unipolar depression,” explains Dr. Amer Burhan, local site lead, researcher at Lawson and neuropsychiatrist at St. Joseph’s Health Care London. “For the first time, we’re studying how effective the treatment is for depression as a result of bipolar disorder and whether it can reduce the risk of cognitive side effects associated with electroconvulsive therapy.”
The clinical trial is being led by the Centre for Addiction and Mental Health (CAMH) and will also be offered through Lawson and University of British Columbia (UBC) Hospital. Lawson researchers will invite eligible patients with treatment-resistant depression from bipolar disorder to participate in the trial at Parkwood Institute, a part of St. Joseph’s Health Care London.
Eligible patients will be randomized to receive either ECT or MST. Patients will be offered support throughout the study with the goal of improving patient outcomes in both groups. Patient outcomes will be compared to study the efficacy of MST and whether it is associated with reduced cognitive side effects.
MST will be delivered under anesthesia in 12 to 20 sessions. Sessions will last 10 to 15 minutes each with 60 to 90 minutes of recovery time.
“Magnetic seizure therapy holds promise of one day replacing electroconvulsive therapy as the gold standard for treatment-resistant depression,” says Dr. Burhan. If proven as a viable first-line treatment, MST would be very easy to implement in existing ECT clinics. MST would therefore be readily available to patients in need.
“We are on the leading edge of the field of brain stimulation for treatment-resistant depression in collaboration with CAMH and UBC,” says Dr. Burhan. “Our goal is to continue informing the care process through clinically-relevant research that serves patients, medical professionals and the public.”
-30-
Lawson Health Research Institute is one of Canada’s top hospital-based research institutes, tackling the most pressing challenges in health care. As the research institute of London Health Sciences Centre and St. Joseph’s Health Care London, our innovation happens where care is delivered. Lawson research teams are at the leading-edge of science with the goal of improving health and the delivery of care for patients. Working in partnership with Western University, our researchers are encouraged to pursue their curiosity, collaborate often and share their discoveries widely. Research conducted through Lawson makes a difference in the lives of patients, families and communities around the world. To learn more, visit www.lawsonresearch.ca.
Senior Media Relations Consultant
Communications & Public Engagement
T: 519-685-8500 ext. 73502
Celine.zadorsky@lhsc.on.ca
Clinical trial will evaluate new therapy for treatment-resistant depression as a result of bipolar disorder
Treatment-resistant depression is a severe form of illness that does not respond to traditional therapies like medication and counselling. It is particularly common in patients with depression from bipolar disorder who are left with limited treatment options.
“There are some mental illnesses that can become resistant to therapy, similar to how infections, for example, can become resistant to antibiotics,” explains Dr. Amer Burhan, researcher at Lawson Health Research Institute and neuropsychiatrist at St. Joseph’s Health Care London. “Patients with those illnesses need more options.”
Brain stimulation is a field that holds promise for this patient population.
“When people are in a state of depression, research shows their brain networks are not functioning properly,” says Dr. Burhan. “Brain stimulation aims to stimulate neurons in the brain to correct activity and improve patient outcomes.”
Through involvement in a national clinical trial, Dr. Burhan and his research team at Lawson are offering new hope with a treatment called magnetic seizure therapy (MST). The study is the first randomized controlled trial to examine the efficacy of MST for patients with treatment-resistant depression as a result of bipolar disorder.
For years electroconvulsive therapy (ECT), one form of brain stimulation, has been the gold standard for patients with treatment-resistant depression. ECT uses an electric field to induce a seizure that provides a therapeutic benefit by stimulating the brain. But while ECT is effective, many patients opt out of treatment due to stigma surrounding the therapy and its potential for cognitive side effects like disorientation and amnesia.
MST has emerged as a promising alternative. MST works in a similar way to ECT but uses a focused magnetic field as opposed to electricity. As a result, MST induces a more focused seizure to reduce the risk of cognitive side effects.
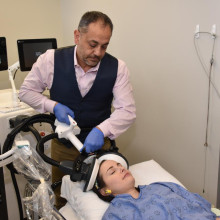
“Magnetic seizure therapy has already been shown as a promising treatment for major depressive disorder or unipolar depression,” explains Dr. Amer Burhan, local site lead for the clinical trial. “For the first time, we’re studying how effective the treatment is for depression as a result of bipolar disorder and whether it can reduce the risk of cognitive side effects associated with electroconvulsive therapy.”
The clinical trial is being led by the Centre for Addiction and Mental Health (CAMH) and will also be offered through Lawson and University of British Columbia (UBC) Hospital. Lawson researchers will invite approximately 30 eligible patients with treatment-resistant depression from bipolar disorder to participate in the trial at Parkwood Institute, a part of St. Joseph’s Health Care London.
Eligible patients will be randomized to receive either ECT or MST. Patients will be offered support throughout the study with the goal of improving patient outcomes in both groups. Patient outcomes will be compared to study the whether MST is effective and whether it is associated with reduced cognitive side effects.
MST will be delivered under anesthesia in 12 to 20 sessions. Sessions will last 10 to 15 minutes each with 60 to 90 minutes of recovery time.
“Magnetic seizure therapy holds promise of one day replacing electroconvulsive therapy as the gold standard for treatment-resistant depression, but we need to learn more about where it fits in our toolbox of potential treatments,” says Dr. Burhan.
If proven as a viable first-line treatment, MST would be very easy to implement in existing ECT clinics. MST would therefore be readily available to patients in need.
“We are on the leading edge of the field of brain stimulation for treatment-resistant depression in collaboration with CAMH and UBC,” says Dr. Burhan. “Our goal is to continue informing the care process through clinically-relevant research that serves patients, medical professionals and the public.”
Those who would like more information about the trial can email @email or @email.





