Search
Search
Supporting a rapid research response during the pandemic
LONDON, ON – Hospital-based research is uniquely positioned to tackle the unprecedented COVID-19 challenge and quickly translate results into illness prevention and care for patients. Lawson Health Research Institute, the research institute of London Health Sciences Centre (LHSC) and St. Joseph’s Health Care London, has awarded more than $202,000 to its research teams to support critical discoveries during the pandemic.
Over 14 projects ranging from improved diagnostics to understanding the impact of the COVID-19 pandemic on frontline health-care workers have been funded through Lawson’s Internal Research Fund (IRF) COVID-19 Pandemic Response Competition.
“Lawson researchers are an intrinsic part of health care in London. Our research community has mobilized quickly to the COVID-19 pandemic and has come forth with innovative ideas to prevent infections, aid recovery and reduce the likelihood of mortality in those most seriously affected,” says Dr. David Hill, Scientific Director for Lawson. “Lawson has reallocated internal granting resources to enable some of the most exciting and credible ideas to be explored.”
Launched in March, this rapid response competition was focused on funding projects, such as two described below, that could impact the management of the pandemic and enhance our understanding of the novel coronavirus.
Using genetic therapy to block entry of the virus
Progression of COVID-19 depends on human proteins that the virus uses to escort itself into lung cells to cause disease. “There are three such ‘virus entry’ proteins that are particularly important for infection. We see them as potential targets for drugs to prevent and treat COVID-19,” explains Dr. James Koropatnick, Lawson Scientist and Principal Investigator for one of the funded studies.
There could be dangers in targeting these proteins, particularly for patients taking certain blood pressure-reducing drugs. The team is studying the safe use of a new type of drug – powerful “antisense” agents that block messenger RNAs which are the biological “blueprints” for the target virus entry proteins – to stop the virus from using those proteins to infect lung cells. They are testing two types of antisense molecules that reduce the amount of the virus entry proteins. “With our existing knowledge and infrastructure in place, this testing could rapidly lead to new, non-vaccine agents to prevent and treat COVID-19, as well as future coronaviruses with pandemic potential,” adds Dr. Koropatnick.
The impact of the pandemic response for those with disabilities
The COVID-19 pandemic has disrupted the daily lives of Canadians, with worsening mental health as people adapt to different stressors. “While many are struggling with this new normal, the degree of disruption resulting from the pandemic presents unique challenges for those with spinal cord injury and brain injury,” says Dr. Eldon Loh, Lawson Associate Scientist also leading one of the funded studies.
Relatively simple tasks such as frequent hand washing can be challenging for those with disability, and they may be more vulnerable to poor outcomes because of the decreased respiratory function many already face. The research team will document and evaluate the effect of the pandemic on the physical and mental health of those with spinal cord and brain injuries. “We plan to use our findings to provide guidance to those living with spinal cord and brain injury, and improve their quality of life during this challenging time. We expect that this study will not only help those with spinal cord and brain injuries, but also people living with other disabilities as well,” notes Dr. Loh. He is also a Physical Medicine and Rehabilitation Specialist at St. Joseph’s.
The IRF competition is supported with funding from clinical departments at LHSC and St. Joseph’s, and from London Health Sciences Foundation and St. Joseph’s Health Care Foundation.
-30-
Lawson Health Research Institute is one of Canada’s top hospital-based research institutes, tackling the most pressing challenges in health care. As the research institute of London Health Sciences Centre and St. Joseph’s Health Care London, our innovation happens where care is delivered. Lawson research teams are at the leading-edge of science with the goal of improving health and the delivery of care for patients. Working in partnership with Western University, our researchers are encouraged to pursue their curiosity, collaborate often and share their discoveries widely. Research conducted through Lawson makes a difference in the lives of patients, families and communities around the world. To learn more, visit www.lawsonresearch.ca.
Senior Media Relations Consultant
Communications & Public Engagement
T: 519-685-8500 ext. 73502
Celine.zadorsky@lhsc.on.ca
Supporting a rapid research response during the pandemic
Hospital-based research is uniquely positioned to tackle the unprecedented COVID-19 challenge and quickly translate results into illness prevention and care for patients. Lawson Health Research Institute, the research institute of London Health Sciences Centre (LHSC) and St. Joseph’s Health Care London, has awarded more than $202,000 to its research teams to support critical discoveries during the pandemic.
Over 14 projects ranging from improved diagnostics to understanding the impact of the COVID-19 pandemic on frontline health-care workers have been funded through Lawson’s Internal Research Fund (IRF) COVID-19 Pandemic Response Competition.
“Lawson researchers are an intrinsic part of health care in London. Our research community has mobilized quickly to the COVID-19 pandemic and has come forth with innovative ideas to prevent infections, aid recovery and reduce the likelihood of mortality in those most seriously affected,” says Dr. David Hill, Scientific Director for Lawson. “Lawson has reallocated internal granting resources to enable some of the most exciting and credible ideas to be explored.”
Launched in March, this rapid response competition was focused on funding projects, such as two described below, that could impact the management of the pandemic and enhance our understanding of the novel coronavirus.
Using genetic therapy to block entry of the virus
Progression of COVID-19 depends on human proteins that the virus uses to escort itself into lung cells to cause disease. “There are three such ‘virus entry’ proteins that are particularly important for infection. We see them as potential targets for drugs to prevent and treat COVID-19,” explains Dr. James Koropatnick, Lawson Scientist and Principal Investigator for one of the funded studies.
There could be dangers in targeting these proteins, particularly for patients taking certain blood pressure-reducing drugs. The team is studying the safe use of a new type of drug – powerful “antisense” agents that block messenger RNAs which are the biological “blueprints” for the target virus entry proteins – to stop the virus from using those proteins to infect lung cells. They are testing two types of antisense molecules that reduce the amount of the virus entry proteins. “With our existing knowledge and infrastructure in place, this testing could rapidly lead to new, non-vaccine agents to prevent and treat COVID-19, as well as future coronaviruses with pandemic potential,” adds Dr. Koropatnick.
The impact of the pandemic response for those with disabilities
The COVID-19 pandemic has disrupted the daily lives of Canadians, with worsening mental health as people adapt to different stressors. “While many are struggling with this new normal, the degree of disruption resulting from the pandemic presents unique challenges for those with spinal cord injury and brain injury,” says Dr. Eldon Loh, Lawson Associate Scientist also leading one of the funded studies.
Relatively simple tasks such as frequent hand washing can be challenging for those with disability, and they may be more vulnerable to poor outcomes because of the decreased respiratory function many already face. The research team will document and evaluate the effect of the pandemic on the physical and mental health of those with spinal cord and brain injuries. “We plan to use our findings to provide guidance to those living with spinal cord and brain injury, and improve their quality of life during this challenging time. We expect that this study will not only help those with spinal cord and brain injuries, but also people living with other disabilities as well,” notes Dr. Loh. He is also a Physical Medicine and Rehabilitation Specialist at St. Joseph’s.
The IRF competition is supported with funding from clinical departments at LHSC and St. Joseph’s, and from London Health Sciences Foundation and St. Joseph’s Health Care Foundation.
Congratulations to all of the researchers and their teams who have been awarded funding:
- Dr. Ian Ball - Predictors of clinical deterioration in hospitalized patients with COVID-19: The CORAL study
- Dr. Doug Fraser - Translational Research Centre: Repository of biological specimens from patients
- Dr. Sean Gill - Rapid identification of therapeutic targets mediating pulmonary microvascular endothelial cell dysfunction in COVID-19 patients
- Dr. Marnin Heisel - Testing Online Meaning-Centered Men’s Groups to Promote Psychological Well-Being and Reduce Despair in the Face of the COVID-19 Pandemic
- Dr. Kimia Honarmand - The impact of the COVID-19 pandemic on frontline healthcare workers: A Southwest Ontario survey
- Dr. Michael Knauer - Improving diagnostic and epidemiologic detection of SARS-CoV-2
- Dr. Jim Koropatnick - Targeting the SARS-CoV-2 receptor with candidate antisense drugs to prevent infection and disease progression
- Dr. Eldon Loh - COVID-19 and Disability: The Impact of a Societal Lockdown on those with Spinal Cord and Brain Injuries
- Dr. Len Luyt - Stapled Peptides as SARS-CoV-2 Fusion Inhibitors
- Dr. Claudio Martin - COVIDOPATHY – An observational study of coagulation abnormalities in patients with COVID-19
- Dr. Tianqing Peng - Developing new ssDNA aptamers targeting S protein receptor binding domain of SARS-CoV-2 to disrupt virus infection cycle as a potential therapy
- Dr. Eva Turley - Hyaluronan signaling through the inflammasome is a target for blunting acute respiratory distress syndrome (ARDS) in COVID-19 patients
- Dr. Ruud Veldhuizen - Next generation exogenous surfactants for COVID-19 and future respiratory pandemics
- Dr. Blayne Welk - Characterizing the burden of surgical care in COVID-19 positive patients
Surgeries take 22 per cent longer in teaching hospitals: Ontario study
Patients undergoing common surgeries in teaching hospitals experience significantly longer surgical times, with durations that are 22 per cent longer on average in these academic centres. This is among the findings of a new study from the Institute for Clinical Evaluative Sciences (ICES) and the Lawson Health Research Institute that was published today in theCanadian Journal of Surgery.
“The ability to train new surgeons is undeniably an essential part of a sustainable healthcare system,” says Christopher Vinden, the study’s lead author who is an adjunct scientist at ICES and a surgeon at the London Health Sciences Centre. “However, we see that surgical instruction significantly lengthens the duration of a range of common procedures, raising important questions such as how to minimize patient risk during teaching procedures, and how to address Ontario funding models that currently don’t adjust for academic versus non-academic centres.”
The researchers examined anonymized records for all adult residents of Ontario who underwent any of 14 common surgical procedures from 2002 to 2012. Of the more than 700,000 surgeries looked at, 21 per cent were performed in a teaching hospital. Previous research has shown that teaching can be associated with longer surgeries, but the authors say this is the first study to examine the magnitude of this increase at a population level, and the first to adjust for patient, procedure and surgeon-related factors such as the experience of the attending surgeon.
They found that of the 14 procedures, hip and knee replacements were least impacted by teaching status, respectively taking 8 and 9 per cent longer (less than 10 minutes longer) in academic centres. However, for the more complex laparoscopic right hemicolectomy (removing the right side of the bowel, usually due to colon cancer) the procedure took 33 per cent longer (62 minutes longer) in teaching hospitals.
Overall, every procedure took longer in teaching hospitals, at a mean of 22 per cent longer duration.
To choose which procedures to investigate, the authors consulted a panel of experts who selected representative procedures that are common in both teaching and community settings. Procedures included laparoscopic cholecystectomy, right hemicolectomy, appendectomy, unilateral inguinal hernia repair, hysterectomy, hip hemiarthroplasty, open reduction and internal fixation for hip fracture, hip and knee arthroplasty, and tonsillectomy. Where applicable, they considered open and laparoscopic procedures separately.
The study authors note that academic centres tend to use dedicated surgical teams for hip and knee replacements, compared to community hospitals where these procedures are typically performed by general orthopedic surgeons in non-dedicated units. They say it’s likely that this team-based specialist approach is helping to shave off that excess teaching time for knee and hip surgeries, and suggest that it may be useful to explore whether these efficiencies can be achieved for other common surgeries in teaching hospitals by adopting specialized units.
Noting that longer surgical durations have been linked to negative patient outcomes, Vinden adds: “In addition to addressing funding gaps created by longer procedure durations at teaching hospitals, it will be vitally important to identify at what point, for each type of surgery, this longer duration due to teaching introduces excess patient risk, and to find ways to minimize this risk. Training the next generation of surgeons should not negatively impact the funding of a teaching hospital, or the health of our patients.”
The study “Teaching surgery takes time: the impact of surgical education on time in the operating room” was published today in the Canadian Journal of Surgery.
Author block: Christopher Vinden, Richard Malthaner, Jacob McGee, J Andrew McClure, Jennifer Winick-Ng, Kuan Liu, Danielle M Nash, Blayne Welk, Luc Dubois.
Lawson Health Research Institute is one of Canada’s top hospital-based research institutes, tackling the most pressing challenges in health care. As the research institute of London Health Sciences Centre and St. Joseph’s Health Care London, our innovation happens where care is delivered. Lawson research teams are at the leading-edge of science with the goal of improving health and the delivery of care for patients. Working in partnership with Western University, our researchers are encouraged to pursue their curiosity, collaborate often and share their discoveries widely. Research conducted through Lawson makes a difference in the lives of patients, families and communities around the world. To learn more, visit www.lawsonresearch.ca.
The Institute for Clinical Evaluative Sciences (ICES) is an independent, non-profit organization that uses population-based health information to produce knowledge on a broad range of health care issues. Our unbiased evidence provides measures of health system performance, a clearer understanding of the shifting health care needs of Ontarians, and a stimulus for discussion of practical solutions to optimize scarce resources. ICES knowledge is highly regarded in Canada and abroad, and is widely used by government, hospitals, planners, and practitioners to make decisions about care delivery and to develop policy. For the latest ICES news, follow us on Twitter: @ICESOntario
Senior Media Relations Consultant
Communications & Public Engagement
T: 519-685-8500 ext. 73502
Celine.zadorsky@lhsc.on.ca
Synthetic surfactant could ease breathing for patients with lung disease and injury
Human lungs are coated with a substance called surfactant which allows us to breathe easily. When lung surfactant is missing or depleted, which can happen with premature birth or lung injury, breathing becomes difficult. In a collaborative study between Lawson Health Research Institute and Stanford University, scientists have developed and tested a new synthetic surfactant that could lead to improved treatments for lung disease and injury.
Lung surfactant is made up of lipids and proteins which help lower tension on the lung’s surface, reducing the amount of effort needed to take a breath. The proteins, called surfactant-associated proteins, are very difficult to create in a laboratory and so the surfactant most commonly used in medicine is obtained from animal lungs.
London, Ontario has a rich legacy in surfactant research and innovation. Dr. Fred Possmayer, a scientist at Lawson and Western University, pioneered the technique used to purify and sterilize lung surfactant extracted from cows. Called bovine lipid extract surfactant (BLES), the therapeutic is made in London, Ontario and used by nearly all neonatal intensive care units in Canada to treat premature babies with respiratory distress.
“When we look at treating adults, surfactant therapy is more difficult. For example, their lungs are 20 times bigger than those of babies and so we need much higher doses of surfactant,” explains Dr. Ruud Veldhuizen, a scientist at Lawson and an associate professor at Western University’s Schulich School of Medicine & Dentistry. “We therefore need to find novel approaches to surfactant therapy for adult patients.”
In this collaborative study, the research team took a new approach to creating synthetic surfactant. Rather than trying to recreate surfactant-associated proteins in the lab, scientists at Stanford created protein mimics. Pioneered by Dr. Annelise Barron, associate professor at Stanford, these protein mimics look like surfactant-associated proteins and have similar properties but are easier to create and more stable. As a result, the team was able to create a new synthetic surfactant.
Collaborating with the Stanford team, Dr. Veldhuizen evaluated the synthetic surfactant in animal models in his research lab at St. Joseph’s Health Care London. The study showed that, unlike other synthetic surfactants currently on the market, the new surfactant equaled or outperformed the animal-derived surfactant in every outcome. This included outperforming animal-derived surfactant in oxygenating blood, which is the lungs’ main purpose.
“The unique ability of the Veldhuizen lab to perform these rigorous and sophisticated studies was a critical aspect of the success of this project,” says Dr. Barron.
“These are very promising results,” says Dr. Veldhuizen. “For the first time, a synthetic surfactant has been developed which appears to be just as effective, if not more so, as that taken from the lungs of animals.”
The team estimates that the synthetic surfactant could be produced at as low as one quarter of the cost of the animal-derived surfactant. With a lower cost the synthetic surfactant could be tested with more lung diseases and injuries in adults and made available in more developing countries.
The team hopes to continue their research with further testing of the synthetic surfactant, including its long term effects. The team also hopes to test its ability to be customized for specific diseases. “Since it is made in the lab, we could combine the surfactant with other drugs like antibacterial agents and deliver it to specific areas of the lung, such as those where an infection is located,” explains Dr. Veldhuizen.
One disease the scientists would like to further study is acute respiratory distress syndrome (ARDS). ARDS is characterized by a low amount of oxygen in the blood due to difficulty breathing. While current surfactants have been tested with ARDS patients, they have not been effective. Dr. Veldhuizen wants to combine this new synthetic surfactant with anti-inflammatory agents and antibacterial agents to test whether patient outcomes are improved.
The study, “Effective in vivo treatment of acute lung injury with helical, amphipathic peptoid mimics of pulmonary surfactant proteins,” is published in Scientific Reports.
Tastings
Attend one of London's premier social dining experiences and celebrate 10 years of sipping world wines, savouring local dishes and supporting London Health Sciences Centre (LHSC).
Date: Thursday, May 5, 2016
Time: 6:00 p.m.
Location: London Hunt & Country Club, 1431 Oxford Street West, London
Attire: Formal, Black-Tie
Tickets: $275 per person
Contact: 519.685.8433, tickets@lhsf.ca or lhsf.ca/tastings
This year's proceeds will make a difference in the lives of cardiac patients and families at LHSC.
Team players: FMT and microbiome research could have widespread impact
There is still much to learn about the human microbiome and its role in fighting disease, but ongoing studies at Lawson Health Research Institute, including a focus on fecal microbial transplants (FMT), are making strides in harnessing this complex system.
FMT is being studied in connection with conditions as varied as non-alcoholic fatty liver disease, rheumatoid arthritis, atherosclerosis, HIV, cancer and multiple sclerosis.
FMT is already in clinical use for the treatment of C. diff (Clostridium difficile), and in addition to showing promise in the treatment of other diseases, it is also being studied as a way to improve response to existing cancer treatments and ease treatment side effects.
Dr. Saman Maleki, a Scientist at Lawson Health Research Institute and the London Regional Cancer Program (LRCP) at London Health Sciences Centre (LHSC), says they’ve just begun to explore the possibilities.
"We are just starting to study FMT as an intervention outside its traditional use in patients with C. difficile infection, and we will be expanding to other areas, particularly in cancer.”
FMT can overhaul a patient’s microbiome, Dr. Maleki explains, and a healthy microbiome is beneficial especially when a treatment is trying to activate the body’s immune system.
Dr. Michael Silverman, Lawson Associate Scientist and Medical Director of St. Joseph’s Health Care London’s Infectious Diseases Care Program, is a pioneer in the field of FMT. He has been performing the procedure since 2003 with C. difficile patients and was one of the first in North America to do so. He sees a wide range of possible applications.
“FMT has enormous potential in being an important adjunctive therapy in many cancers. It may, for example, help cancer patients respond to immunotherapy,” says Dr. Silverman, who is also Chair/Chief of Infectious Disease at St. Joseph’s Health Care London, LHSC and Western University. “The potential to impact autoimmune and metabolic diseases is also quite exciting, but still in early development.”
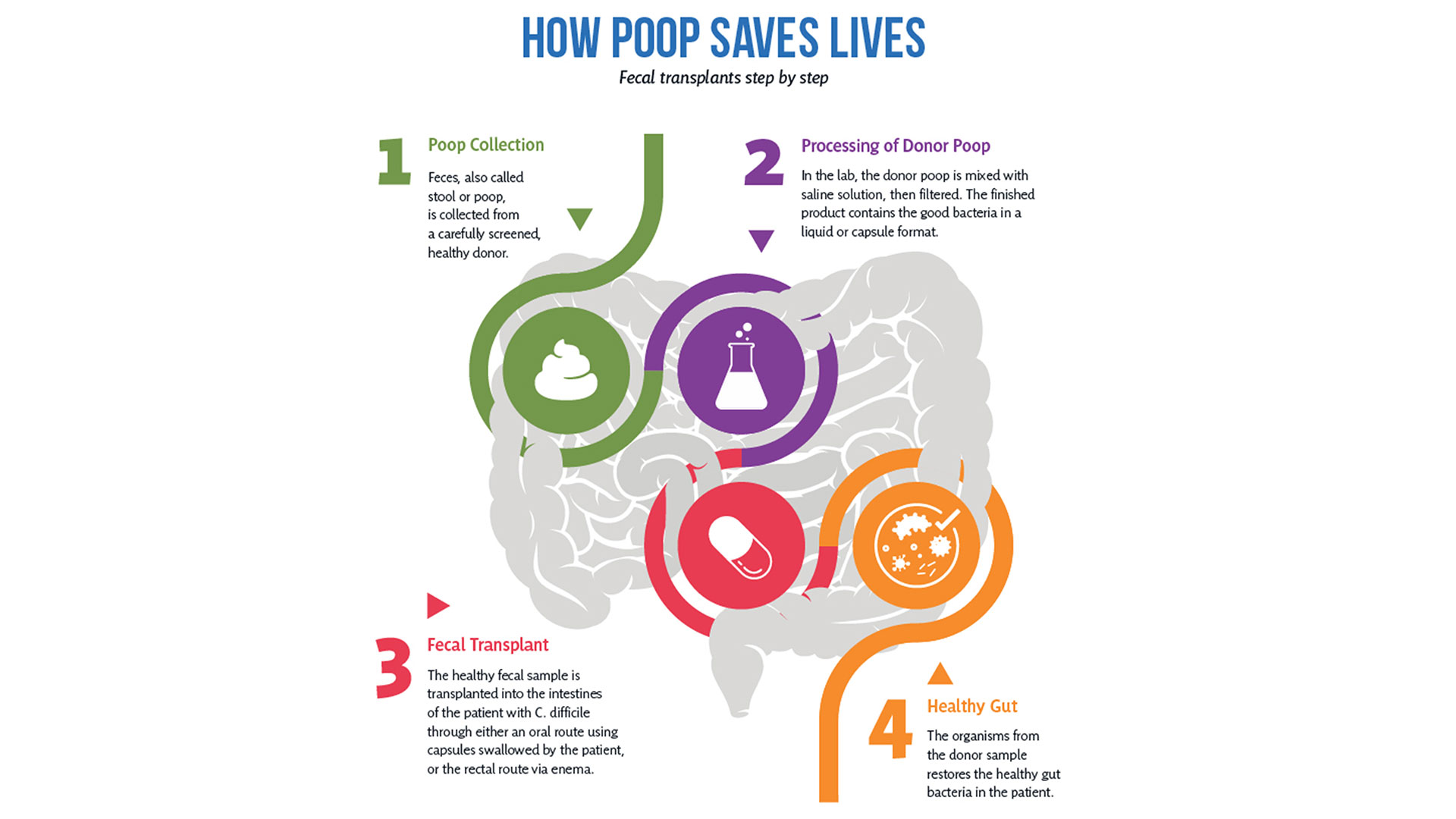
So how does it work?
After rigorous screening, stool from a healthy donor is collected and then processed in a lab into a liquid or capsule containing the good bacteria, which can then be administered to a patient’s gastrointestinal tract.
The Lawson team is also one of few delivering FMT using specially-prepared oral capsules. Introduced in 2018, they have been a game changer in patient acceptance and ease of administration, according to Research Coordinator Dr. SeemaNair Parvathy, who has been coordinating the program since 2015.
“There is a link between the fitness of the intestinal microbiome and the fitness of the immune system,” says Dr. John Lenehan, Associate Scientist at Lawson and Medical Oncologist at LHSC. “A ‘healthy’ microbiome leads to a more robust immune response when using immunotherapy. FMT from a healthy donor is expected to improve the fitness of the recipient’s intestinal microbiome and promote a better immune response.”
People with chronic disease can often experience what’s called a ‘leaky gut,’ allowing food, bacterial and microbial components to pass through the intestinal wall, negatively impacting the immune system.
“When people get FMTs their intestinal permeability improves – meaning it actually reduces,” says Dr. Jeremy Burton, Lawson Scientist and Research Chair of Human Microbiome and Probiotics at Lawson and St. Joseph’s. “What changes that intestinal permeability? The microbes at the site. They play a role in interacting with the host cells, providing nutrients and vitamins.”
With the immune system so closely tied to the health of the microbiome, it’s not surprising scientists are exploring how strengthening one can have a big impact on the other.
Boosting immunotherapy
Immunotherapy can be used to either stimulate or suppress the immune system to help the body fight disease, and FMT is showing promise in reducing resistance to the treatment.
While immunotherapy has been effective in treating a number of cancers – the number one cause of death in Canada – not all patients respond to the treatment.
But early work presented at a conference by the Lawson team for the Journal for ImmunoTherapy of Cancer has shown that using FMT to modify the microbiome could reduce resistance to immunotherapy. The study involved patients from LHSC with advanced melanoma, a type of skin cancer.
While in the very early stages, the combination of FMT and anti-PD1 immunotherapy has been found to be safe, and it appears that FMT could make tumours more responsive to the immunotherapy treatment.
“Microbiome-based treatment strategies, including FMT, have a high potential in oncology,” says Dr. Maleki. “Our team is also exploring its potential in treating pancreatic cancer.”
The research is so interesting that a recent Nature article listed the Phase I melanoma trial as “seminal” research. The study was also unique in that it used healthy donors, as opposed to donors who had previously responded to immunotherapy treatment.
A separate Lawson study with LHSC patients with metastatic renal cell carcinoma, a type of kidney cancer, published in the Journal of Clinical Oncology, also looked at combining immunotherapy and FMT to ease the adverse effects of the treatment.
The Phase I study, led by Dr. Maleki and Dr. Ricardo Fernandes, Medical Oncologist at LHSC, found adding FMT to doublet immunotherapy was safe, but further study is needed to determine whether it could bring about changes in the microbiome and immune system.
Dr. Lenehan says Lawson researchers are in a position to be leaders in this field in the near future for two reasons.
“One is that other academic researchers have not been able to assemble the expertise, and some who have, do not have the access to healthy donor stool. The second is that some biotechnology companies are interested in FMT, but almost exclusively for C. difficile infections.”
Autoimmune, metabolic and other illnesses
Two other areas that have seen recent advances include FMT for the treatment of non-alcoholic fatty liver disease (NAFLD) and multiple sclerosis.
“The gut microbiome is very important in the metabolism of foods and metabolic products. It can therefore have a major effect on obesity and atherosclerosis,” says Dr. Silverman. “It also is tightly involved in regulating the immune system and therefore moderating the microbiome may potentially impact autoimmune diseases.”
A study published in 2020 by the team in The American Journal of Gastroenterology showed that FMT appears to reduce intestinal permeability in patients with NAFLD.
The number of people with NAFLD is growing rapidly and studies show patients have different microbiota than healthy persons.
The trial included 21 NAFLD patients from LHSC and St. Joseph’s. While the researchers found no changes in percentage of liver fat or insulin resistance, they observed significant reduction in intestinal permeability in those patients who had elevated intestinal permeability at the study’s start (seven patients in total). They also observed changes to the gut microbiome in all patients who received a fecal transplant from a healthy donor.
“Metabolic syndromes including obesity and its complications of NAFLD and atherosclerosis are massive public health problems. Any impact on these would be of huge importance,” Dr. Silverman adds. “Autoimmune diseases also cause major morbidity and mortality. We have a lot of work to do before we can consider FMT as a routine therapy for any of these conditions, but the long-term promise is great.”
Research into the use of FMT for treatment of patients with multiple sclerosis is in the very early stages. But patients with MS show a difference in gut microbiota and higher small intestine permeability, which could contribute to the development of the disease.
A Phase I trial by the Lawson team published in the Multiple Sclerosis Journal – Experimental, Translational and Clinical, found FMT to be safe and tolerable.
While the study was very small, MS patients treated with FMT were found to have beneficial changes to gut microbiota and intestinal permeability, but further study is needed to determine if FMT could be used as a treatment.
Lawson scientists are also currently studying the use of FMT for patients with atherosclerosis, along with ongoing studies on melanoma and lung cancer. Funding for a study on pancreatic cancer has been secured and researchers are in the process of planning trials for a number of other applications.
Dr. Lenehan says, “The microbiome is connected to several diseases and their treatments. Evidence is growing that an individual’s health is related to their microbiome.”

The donor challenge
The challenge of finding fecal donors for FMT and the cost of that process remain an issue for research into this promising treatment, even as more potential applications are discovered.
There is currently no process in place to match donors and recipients – as with organ donation – but there is an extensive screening process for both infectious and non-infectious conditions, one that needs to be repeated if a donor experiences any lifestyle changes.
Dr. Burton says, “We still don't understand the full role of the microbiota. We have to ensure that we're not giving patients a microbiota that might cause them some other issue in the future, so the donors are screened very thoroughly for that.”
Screening also excludes donors with an increased risk of developing the diseases scientists are hoping to treat, such as metabolic syndrome related conditions.
A 2017 article published in Open Forum Infectious Diseases by Drs. Silverman and Burton found the cost of screening high numbers of potential donors could make establishing local programs extremely difficult, so having a central program such as the one in London could help patients in many regions.
In the study, only five of 46 potential donors passed the history, examination, blood, stool and urine tests, and of the five, four later travelled or had illnesses that made donation inadvisable.
The search continues in London for potential donors to help drive this research forward. You can read one donor’s story and learn how you can get involved here.
More on FMT and the microbiome:
Drugs vs. bugs: Harnessing the microbiome to improve treatments
Communications Consultant & External Relations
Lawson Health Research Institute
T: 519-685-8500 ext. ext. 64059
C: 226-919-4748
@email
Third clinical trial launched to study whether type 2 diabetes can go into remission
Since launching two years ago, an innovative study that aims to induce remission of type 2 diabetes has captured the attention of hundreds of Londoners. For those with type 2 diabetes like Greg Ackland and Jocelyne Chauvin, the idea of stopping all medications has translated from a dream to a reality through participation in the REMIT study at Lawson Health Research Institute (Lawson).
With a family history of type 2 diabetes, Greg Ackland was first diagnosed over six years ago when he underwent an operation for a hernia. He developed a mild infection and, while being treated, his care team discovered his blood sugar levels were high.
Ackland started treatment and was eventually taking four pills per day. “I watched the progression of my medications and thought ‘I’m losing this battle,’” says 51 year-old Ackland.

Above: Greg Ackland, research participant in the REMIT study
He saw information about the REMIT study on the local news and after meeting the criteria he was enrolled. The outcomes have been incredible. Ackland has now stopped all medications and his blood sugar levels are good. He has recommitted to exercise which has resulted in weight loss and muscle gain.
“I’m glad I jumped at the opportunity to participate in this trial,” says Ackland. “I’ve reset myself.”
Lawson is one of seven Canadian sites taking part in the REMIT study, which is considered a significant departure in strategy in the care of people with type 2 diabetes. The study consists of a series of clinical trials that tests an aggressive approach in recently diagnosed patients. The first two trials in London saw significant interest in participation from those with type 2 diabetes. A third REMIT trial is now being launched providing another opportunity for individuals with type 2 diabetes to take part.
“The goal of the REMIT study is to take a proactive approach to help people early in the disease, normalize their blood sugars for a period of 12 weeks and then slow the progression of the disease and the need for additional medications,” says Dr. Irene Hramiak, Lawson researcher, endocrinologist, and Chief of the Centre for Diabetes, Endocrinology and Metabolism at St. Joseph’s Health Care London. “We want to know if we can induce remission, for how long and whether it matters what combination of medications we use.”
The standard treatment for people with type 2 diabetes is to start on a single medication, which is then followed by the addition of more drugs and insulin as the disease progresses. In the REMIT study, patients receive intensive treatment early in their care journey that consists of two diabetes medications plus insulin at bedtime for three months to see if remission can be induced. In addition, patients are supported to make lifestyle changes with a diet and 30 minutes of exercise each day.
“When I saw a gentleman on the news talking about the REMIT study and how it allowed him to stop taking all medications, I thought ‘cool!’” says Jocelyne Chauvin. The 62 year-old Londoner was first diagnosed with type 2 diabetes three years ago after a regular checkup. While it was difficult news, she had a family history of the disease and health issues before her diagnosis.

Above: Jocelyne Chauvin, research participant in the REMIT study
“I started taking one pill a day and was told I’d be up to four pills a day within six months,” says Chauvin. “But with good nutrition and exercise I worked hard to stay on one pill a day for three years.”
After hearing about the study, she contacted the research team and, after meeting the criteria, was enrolled in April 2017. Chauvin has now stopped all medications and says she feels much better. She exercises more and her blood sugar levels are close to normal.
“This is my first time participating in a clinical trial and I’m very excited about my experience,” says Chauvin.
REMIT is being led by the Population Health Research Institute (PIHR), a joint institute of McMaster University and Hamilton Health Sciences. The study follows a PHRI pilot study of early aggressive treatment that resulted in up to 40 per cent of intervention group participants with type 2 diabetes going into remission and not needing any diabetes treatment for at least three months.
“The idea of putting type 2 diabetes into remission is changing the way we think about the disease. It has a strong appeal to both those with type 2 diabetes and clinicians,” says Dr. Hramiak. “It’s changing the paradigm of when and how to use medication for type 2 diabetes.”
Those who would like more information about the trial can call 519-646-6100 ext. 65373.
This will be a huge benefit for society because sepsis is the leading cause of death worldwide
It’s a discovery that has been more than ten years in the making: the use of a human protein to potentially treat patients with sepsis.
Lawson Scientist Dr. Qingping Feng noticed that a human protein called annexin A5 showed positive results with sepsis back in 2007.

Fast forward 14 years later to now, and this discovery could very well be the first ever viable treatment for sepsis patients, including severe COVID-19 patients who develop sepsis. “With COVID initially, it is in the airway and then in the lungs, then from there the inflammatory response in fact spreads to the whole body,” says Dr. Feng, Ivey Chair in Molecular Technology at Western University's Schulich School of Medicine & Dentistry. “Sepsis causes major organ dysfunction and carries a high mortality unfortunately.”
It has become a challenging issue for Intensive Care Physician at London Health Sciences Centre (LHSC), Dr. Claudio Martin, who can only do so much to treat severe COVID-19 patients that develop sepsis.
“What we have seen is a very primary severe respiratory failure to the lungs for severe COVID patients,” says Dr. Martin, Associate Scientist at Lawson. “We have used steroids and other treatments to try to help, but the results and effects aren’t dramatic and we see patients who have these treatments and still progress and end up in the ICU.”
However, Dr. Feng and his team has found in a pre-clinical study, that annexin A5 can inhibit inflammation, improve organ function, and survival when treating sepsis.
Another potentially deadly situation for COVID-19 patients is cell death and blood clots, specifically near the lungs. The good news is that the research team also believes the annexin A5 drug will prevent these complications through the drugs anti-apoptotic (cell death prevention) and anti-coagulant (blood clot prevention) properties.

Supported by provincial funding through Ontario's COVID-19 Rapid Research Fund, the research team has launched a clinical trial with critically ill COVID-19 patients at LHSC, using a manufactured form of annexin A5. The goal is to enroll a total of 60 patients for the clinical trial, and enrollment has already begun. “Patients are receiving standard treatment and then those enrolled will also receive the annexin,” says Dr. Martin. “It’s a placebo blinded clinical trial, so patients will either get a lower dose of annexin, a higher dose of annexin, or a placebo.”
If the clinical trial shows promising results, Dr. Feng says the team plans on expanding into a larger phase three trial with not just COVID-19 patients with sepsis, but other sepsis patients as well. “If in fact annexin A5 is shown to be effective in sepsis, then this will be a huge benefit for society because sepsis is the leading cause of death worldwide.”
The drug is currently being produced through a partnership with Suzhou Yabao Pharmaceutical R&D Co., Ltd., based in China, Lawson Health Research Institute, and WorldDiscoveries. “Our long-standing partnership with Suzhou Yabao has enabled annexin A5 drug development to proceed to this point,” says Kirk Brown, Manager of Business Development, Lawson Health Research Institute. “We are now in a unique position through this trial to offer a potential life-saving treatment for this emergent global disease, with the objective of soon expanding to all cause septic patients.”