Search
Search
Research encourages re-evaluation of special nerve treatment for chronic pain
LONDON, ON – Hospital researchers from Lawson Health Research Institute have published a recent study that assessed the use of a specialized treatment for chronic pain and its impact on health care use and opioid prescribing.
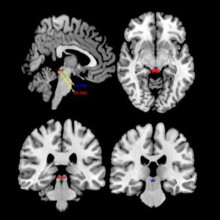
Paravertebral blocks (PVBs) belong to a broader group of procedures called “nerve blocks.” A recent Toronto Star report noted that OHIP has been billed $420 million for nerve block procedures since 2011. PVBs involve injecting medication around the nerves where they exit the bones of the spine, at different locations depending on the patient and the chronic pain they are experiencing.
The regular use of these procedures has been questioned by health care providers due to the high cost and limited evidence of their benefit in reducing chronic pain. While the effectiveness of PVBs has been examined in trauma, cancer pain and regional anesthesia during surgery, they have not been evaluated for use in chronic pain despite widespread use in Ontario.
It is estimated that one in five Canadians live with chronic pain. Pain that persists can affect all aspects of someone’s life and health, particularly when it is not being managed.
This new study from London researchers found that 66,310 patients had a PVB between July 2013 and March 2018, and 47,723 patients were included in the study. In the year after a patient’s first PVB, there was a significant increase in the number of physician visits. Additional PVBs were frequently performed after the first treatment, with over 26 per cent of patients receiving a PVB ten or more times in one year, with almost eight per cent of patients receiving 30 or more. No overall change was found in opioid dosage in the year after PVB was initiated compared to the year before.
“Frequent use of PVB is common. Initiating treatment with PVCs is associated with marked increases in health care utilization, which includes physician visits and other injection procedures,” explains Dr. Eldon Loh, Lawson Associate Scientist and Physiatrist at St. Joseph’s Health Care London.
This research provides a broad perspective on the use of PVBs in Ontario, and on the use of nerve blocking treatments in general. There has been a concern for several years about the over use of these procedures; however, this is the first study to systematically document the impact on health care utilization and opioid use.
"We hope that from this study, the appropriate use of PVBs and other pain interventions will be re-evaluated at a provincial level to ensure the use of health resources is being properly managed and we achieve the best outcome for patients,” Dr. Loh adds.
The study, “A Retrospective Cohort Study of Healthcare Utilization Associated with Paravertebral Blocks for Chronic Pain Management in Ontario,” is published in the Canadian Journal of Pain.
-30-
Lawson Health Research Institute is one of Canada’s top hospital-based research institutes, tackling the most pressing challenges in health care. As the research institute of London Health Sciences Centre and St. Joseph’s Health Care London, our innovation happens where care is delivered. Lawson research teams are at the leading-edge of science with the goal of improving health and the delivery of care for patients. Working in partnership with Western University, our researchers are encouraged to pursue their curiosity, collaborate often and share their discoveries widely. Research conducted through Lawson makes a difference in the lives of patients, families and communities around the world. To learn more, visit www.lawsonresearch.ca.
Senior Media Relations Consultant
Communications & Public Engagement
T: 519-685-8500 ext. 73502
Celine.zadorsky@lhsc.on.ca
Research encourages re-evaluation of special nerve treatment for chronic pain
Hospital researchers from Lawson Health Research Institute have published a recent study that assessed the use of a specialized treatment for chronic pain and its impact on health care use and opioid prescribing.
Paravertebral blocks (PVBs) belong to a broader group of procedures called “nerve blocks.” A recent Toronto Star report noted that OHIP has been billed $420 million for nerve block procedures since 2011. PVBs involve injecting medication around the nerves where they exit the bones of the spine, at different locations depending on the patient and the chronic pain they are experiencing.
The regular use of these procedures has been questioned by health care providers due to the high cost and limited evidence of their benefit in reducing chronic pain. While the effectiveness of PVBs has been examined in trauma, cancer pain and regional anesthesia during surgery, they have not been evaluated for use in chronic pain despite widespread use in Ontario.
It is estimated that one in five Canadians live with chronic pain. Pain that persists can affect all aspects of someone’s life and health, particularly when it is not being managed.
“Frequent use of PVB is common. Initiating treatment with PVCs is associated with marked increases in health care utilization, which includes physician visits and other injection procedures,” explains Dr. Eldon Loh, Lawson Associate Scientist and Physiatrist at St. Joseph’s Health Care London.
This research provides a broad perspective on the use of PVBs in Ontario, and on the use of nerve blocking treatments in general. There has been a concern for several years about the over use of these procedures; however, this is the first study to systematically document the impact on health care utilization and opioid use.
"We hope that from this study, the appropriate use of PVBs and other pain interventions will be re-evaluated at a provincial level to ensure the use of health resources is being properly managed and we achieve the best outcome for patients,” Dr. Loh adds.
Research shared and celebrated at 17th Annual Mental Health Research Half Day
From falls prevention to depression therapies, scientists at Lawson Health Research Institute are conducting important mental health studies. Held on Thursday, September 15, the 17th Annual Mental Health Research Half Day at the Parkwood Institute Mental Health Program was a chance to share and celebrate this research.
The Mental Health Research Half Day featured poster and oral presentations, as well as the 12th Annual Tony Cerenzia Research Lecture. Clinical, administrative and research staff attended to learn more about research happening at Parkwood Institute and the Southwest Centre for Forensic Mental Health Care, part of the St. Joseph’s Health Care London family.
“The Mental Health Research Half Day provides an opportunity for researchers at Parkwood Institute and the Southwest Centre for Forensic Mental Health Care to present their research findings to clinical and administrative staff,” said Dr. Richard O’Reilly, Director of Psychiatric Research at Parkwood Institute & Southwest Centre and a Scientist at Lawson. “It is important that all clinical staff, who may not be directly involved in research, know what studies are being conducted and their impact on patient care.”
The 12th Annual Tony Cerenzia Research Lecture was delivered by Dr. Nathan Herrmann, Associate Scientist, Evaluative Clinical Sciences, Hurvitz Brain Sciences Research Program, Sunnybrook Research Institute and Head of the Division of Geriatric Psychiatry at Sunnybrook Health Sciences Centre. Dr. Herrmann delivered an engaging lecture titled “Managing Neuropsychiatric Symptoms in Dementia: An Evidence-Based Approach”.
Attendees were engaged not only by this highly informative lecture but also by the poster and oral presentations which covered a broad range of research topics. Presenters were also enthusiastic about the day and the opportunities it provided.
“The Mental Health Research Half Day is a great event which provides networking opportunities here at Parkwood Institute. It allows staff from across St. Joseph’s to learn about different research happening across program areas,” said Erin Finley, an Occupational Therapist (OT) in Geriatric Psychology at Parkwood Institute.
Finley and her colleagues were one of seven poster presentations. Their research project, titled “Fall prevention initiative in geriatric psychiatry”, aimed to reduce the rate of falls with injury among patients with dementia in a behavioural health unit. Within an 18-bed unit, they were able to significantly reduce falls with injury with zero incidences in the last two months of their data collection period.
Researchers are combining new technologies to examine blood proteins in COVID-19 patients
LONDON, ON – Published in the Journal of Cellular and Molecular Medicine, a team at Lawson Health Research Institute have discovered unique patterns of blood plasma proteins in critically ill patients that may help develop a more personalized approach to treating severe COVID-19.
Called the plasma proteome, the proteins being studied are released by cells that often play an important role in the body’s immune response to viruses. The research team studied how they adapt and change to a COVID-19 infection.
As part of the study, blood samples were taken from 30 subjects in three patient groups at London Health Sciences Centre (LHSC). One group had patients with COVID-19, another group had patients with severe infection but were negative for COVID-19, and the third was a healthy control group. Blood samples were drawn on the day of critical care admission and again on days three, seven and ten in hospital.
“We collected plasma from these patients and measured well over a thousand proteins with great accuracy using new technology that combines immunology and genomics,” says Dr. Douglas Fraser, Lawson Scientist, Critical Care Physician at Children’s Hospital at LHSC and Professor at Western University’s Schulich School of Medicine & Dentistry. “With the use of this advanced technology, we were able to better analyze the protein patterns and better understand what is happening with COVID-19, especially in critically ill patients.”
The research team found that COVID-19 patients demonstrated changes in immunosuppression pathways, which typically keeps the immune system balanced. In critically ill patients, the changes were heightened. Analyses of the plasma proteome helped researchers determine which cells in the body are active during the disease state and which signaling pathways were activated.
"In-depth analysis of the human plasma proteome helps us capture tissue proteins that can provide us with information regarding organ integrity during infection," says Dr. Cristiana Iosef, Lawson Research Associate PhD. “This is important because it will allow us to search for new blood biomarkers that are specific for COVID-19 patients.”
This research, which used state-of the-art analysis technology, was enabled by existing expertise and technologies through Children’s Health Research Institute (CHRI), a program of Lawson.
“This study has allowed us to understand the progression of the disease processes in very sick patients, providing us clues on the body’s immune system and other systems that were reacting to the severe disease,” says Dr. Victor Han, Lawson Scientist, Director of CHRI and Professor at Schulich Medicine & Dentistry. “We hope that this knowledge will allow us to identify the patients who will become severely ill and develop new therapies to counteract the changes occurring within their bodies.”
Dr. Fraser, who is also a scientist at CHRI, adds that the team can now examine potential new drug therapies with the hopes of improving outcomes for these patients.
The next steps for the research team will be to use this technology to examine plasma biomarkers in long COVID patients to determine why some develop prolonged disease after a COVID-19 infection.
Lawson Health Research Institute is one of Canada’s top hospital-based research institutes, tackling the most pressing challenges in health care. As the research institute of London Health Sciences Centre and St. Joseph’s Health Care London, our innovation happens where care is delivered. Lawson research teams are at the leading-edge of science with the goal of improving health and the delivery of care for patients. Working in partnership with Western University, our researchers are encouraged to pursue their curiosity, collaborate often and share their discoveries widely. Research conducted through Lawson makes a difference in the lives of patients, families and communities around the world. To learn more, visit www.lawsonresearch.ca.
Senior Media Relations Consultant
Communications & Public Engagement
T: 519-685-8500 ext. 73502
Celine.zadorsky@lhsc.on.ca
Researchers developing photoacoustic hand-held probe for tumour detection during breast conserving surgery
Researchers at Lawson Health Research Institute (Lawson) are developing a hand-held photoacoustic imaging probe to be used during breast conserving surgery to quickly and accurately verify if all cancerous tissue has been removed.
Surgeons currently do not have real-time technology to guide tumour removal during surgery.
Using current tools, there is a 20 per cent chance that cancerous cells will be left behind, risking recurrence and repeat surgery.
Breast cancer represents 25 per cent of all new cancer diagnoses in women and 13 per cent of all cancer related deaths in women. Treatment for breast cancer often requires either complete breast removal in severe cases, or surgical removal of the cancerous tumour in combination with other therapies. Removing only the tumour is called breast conserving surgery.
Image


Photoacoustic Screening
The new device is an extension of the photoacoustic screening (iPAS) technology developed in the laboratory of Dr. Jeffrey Carson, Principal Investigator and Lawson Scientist. The technique uses light and sound to capture 3D images of surgically removed breast tissue. Their studies show that iPAS can catch up to 75 per cent of missed tumour cells, decreasing the odds of failed surgery to five per cent.
Dr. Muriel Brackstone, Associate Scientist at Lawson, Head of the Breast Care Clinic at St. Joseph’s Hospital London, and Surgical Oncologist at London Health Sciences Centre, brings her clinical expertise to the project.
“With the first generation iPAS technology, we would remove the tumour, take it to the lab for imaging and wait to see if there was a rim of normal tissue around the removed tumour so we knew it was removed completely. The wait was anywhere from 20 minutes to an hour. During that time, the patient is under anesthesia, the surgical team is idle and precious OR time is being used,” explains Dr. Brackstone.
A hand-held tool that surgeons can use
Creation of a hand-held probe to be used in the operating room is the next step in the advancement of this new technology. Elina Rascevska, biomedical engineering student at Western University, recently joined the Lawson team to convert lab-based iPAS technology into a hand-held device.
“We have developed a prototype of the iPAS probe, and once we can verify the quality of the images it produces, we will give it to Dr. Brackstone to test in the OR,” says Rascevska.
The iPAS probe does not need a trained operator and would be used by the surgical team. Instead of imaging the removed tissue, it scans the surgical cavity in real time to give the team a faster and more accurate indication as to whether the cancerous tissue has been removed.
“If we can progress this technology to a point where physicians can use it as part of standard protocols, we will have reduced the amount of time each patient needs to spend in the OR, the amount of call-backs and repeat surgeries, and ultimately improve quality of life for patients with breast cancer,” adds Dr. Carson.

(From left): Dr. Jeffrey Carson, Elina Rascevska, Dr. Muriel Brackstone
Researchers investigate a new method of sedation for paediatric patients
Scientists at Children’s Health Research Institute (a program of Lawson Health Research Institute), Sunnybrook Research Institute and The Hospital for Sick Children (SickKids) are working together to study the potential benefits of inhaled sedation as an alternative to keep critically ill children sedated and comfortable.
“Many sick children need support from a ventilator and other life-saving treatments, and may require intravenous (IV) sedatives to tolerate these uncomfortable therapies,” says Dr. Rishi Ganesan, Lawson Associate Scientist and Paediatric Neurocritical Care Physician at Children’s Hospital at London Health Sciences Centre (LHSC). “However, our current sedation options may contribute to a complication called delirium. We are interested in evaluating if delirium and long-term neurological complications are lower in children receiving inhaled sedation compared to those receiving IV sedation, which is the current standard of care.”
Delirium is an acute change in mental state that children in critical care can sometimes develop as a result of their critical illness and the medications and therapies they receive during their hospital stay. Delirium presents as confusion, disorientation, agitation, excessive drowsiness or poor attention. Dr. Marat Slessarev, Lawson Scientist and Critical Care Physician at LHSC, has been researching and comparing inhaled sedation to IV sedation in adults since the pandemic hit in 2020 in a collaborative trial called SAVE-ICU with Dr. Angela Jerath, Anesthesiologist and Scientist at Sunnybrook.
“One of the challenges with IV sedation is that we do not have a way to measure the level of sedatives in the blood,” explains Dr. Slessarev. “Critically ill patients that are sedated can sometimes develop issues with the kidney and liver, which are both important in eliminating the sedatives from the blood stream.”
Through this novel collaborative research, the team is now looking at the potential benefits of inhaled sedation in paediatric patients.
“Inhaled sedatives are an alternative to currently used IV sedatives, and they may reduce delirium and accelerate brain recovery. Inhaled sedatives are used safely every day in operating rooms, widely available and inexpensive,” explains Dr. Jerath. “In contrast to IV sedatives, they do not accumulate in the body, are rapidly eliminated via the lungs, promote faster awakening and discharge from a ventilator, and reduce inflammation – which may be a contributing factor to delirium.”
Enrollment for the ABOVE trial is beginning at Children’s Hospital at LHSC and SickKids. The pilot study will enroll 60 critically ill paediatric patients who will be randomized into two groups; one group will receive inhaled sedation while the other will get standard IV sedation. Once the pilot phase of the trial is complete, the team hopes to expand this trial across the country with more paediatric intensive care units (ICUs) joining the larger trial.
“The field of critical care has made significant strides in life-saving technologies and therapies in recent years, but now we are focused on finding ways to ensure our patients continue to do well after leaving the hospital,” says Dr. Nicole McKinnon, Critical Care Physician and lead investigator at SickKids and a Scientist Track Investigator at SickKids Research Insitute. “This trial is a first step in better understanding the effects of sedative and pain medications on children’s longer-term neurocognitive development. Our research will be key to providing critically ill children with the greatest chance to flourish at home.”
“This has the potential to change how critically ill children are cared for in paediatric ICUs across Canada and the world,” adds Dr. Ganesan. “We hope that inhaled sedation makes a difference in children’s long-term functional outcomes, so they can thrive and achieve their full potential.”
The ABOVE Trial recently received funding through a Canadian Institute for Health Research (CIHR) grant.
About Sunnybrook Research Institute: Sunnybrook Research Institute (SRI) is the research arm of Sunnybrook Health Sciences Centre, an internationally recognized academic health sciences centre fully affiliated with the University of Toronto. With well-established programs in basic and applied sciences which span across three scientific platforms and ten clinical programs, SRI is developing innovations in care for the more than 1.3 million patients the hospital cares for annually. To learn more, visit www.sunnybrook.ca/research
About The Hospital for Sick Children: The Hospital for Sick Children (SickKids) is recognized as one of the world’s foremost paediatric health-care institutions and is Canada’s leading centre dedicated to advancing children’s health through the integration of patient care, research and education. Founded in 1875 and affiliated with the University of Toronto, SickKids is one of Canada’s most research-intensive hospitals and has generated discoveries that have helped children globally. Its mission is to provide the best in complex and specialized family-centred care; pioneer scientific and clinical advancements; share expertise; foster an academic environment that nurtures health-care professionals; and champion an accessible, comprehensive and sustainable child health system. SickKids is a founding member of Kids Health Alliance, a network of partners working to create a high quality, consistent and coordinated approach to paediatric health care that is centred around children, youth and their families. SickKids is proud of its vision for Healthier Children. A Better World.
Lawson Health Research Institute is one of Canada’s top hospital-based research institutes, tackling the most pressing challenges in health care. As the research institute of London Health Sciences Centre and St. Joseph’s Health Care London, our innovation happens where care is delivered. Lawson research teams are at the leading-edge of science with the goal of improving health and the delivery of care for patients. Working in partnership with Western University, our researchers are encouraged to pursue their curiosity, collaborate often and share their discoveries widely. Research conducted through Lawson makes a difference in the lives of patients, families and communities around the world. To learn more, visit www.lawsonresearch.ca.
Communications Consultant & External Relations
Lawson Health Research Institute
T: 519-685-8500 ext. ext. 64059
C: 226-919-4748
@email
Researchers looking to better personalize treatment for PTSD
Post-traumatic stress disorder (PTSD) is a condition that often develops in those who have experienced a traumatic event. PTSD affects close to 10 per cent of Canadians, and in some cases, can be difficult to treat.
In a new study through Lawson Health Research Institute and Western University’s Schulich School of Medicine & Dentistry, scientists are looking at a form of treatment called deep brain reorienting (DBR) for those suffering from PTSD.
“In our study we are looking at ways of trauma processing at the foundation of the stress response,” explains Dr. Ruth Lanius, Associate Scientist at Lawson and Professor at Western’s Schulich School of Medicine & Dentistry. “When you are faced with a stressor the first thing you do is turn your attention to it, and when you do that you engage your head and neck muscles.”

DBR was developed by Scottish psychiatrist Dr. Frank Corrigan and focuses on the muscles of the shoulders, neck, head and face of patients when recalling a traumatic event. This form of trauma processing starts right at the moment the stressor is noticed by the individual, which is often quickly followed by a moment of shock. By addressing the stressor at this level, doctors can target the foundation of where the stress response begins.
“When patients bring up a trigger in their mind, we look at what happens in terms of the tension in their head and neck. We process that gently by asking the patient to notice and stay with that tension without trying to release it, and over time the tension usually decreases,” explains Dr. Lanius, who is also a Psychiatrist at London Health Sciences Centre. “The second part involves addressing the shock response. You turn your attention to the stressor and then notice any moments of shock that follows, which is often associated with tension behind the eyes and in the shoulders. Finally, you address the visceral sensations associated with feelings and emotions associated with the stressor.”
Dr. Lanius believes that by using DBR and focusing on muscle and visceral responses, it will help patients suffering from PTSD to process the visceral feelings associated with the trauma and its triggers.
“After this form of treatment, patients often say they feel safer and don’t see themselves negatively, like they did in the past,” says Dr. Lanius. “This is because we change those intense visceral feelings and it allows the person to reevaluate who they are and how they interact with the world.”
The research team is hoping to recruit approximately 60 people to take part in the DBR study. Participants will undergo a psychological interview and an MRI, then eight sessions of treatments. Following those sessions, participants will have another interview and MRI to assess if any positive changes were made within the neurological networks and/or in psychological systems.
“Our hopes are to really personalize treatment for those living with PTSD,” says Dr. Lanius. “Some people have difficulty tolerating some forms of trauma processing and in my experience this treatment is a gentler approach for those individuals.”
Interested participants who have been diagnosed with PTSD can contact Suzy Southwell, Research Coordinator, at 519-685-8500 ext. 35186 or @email.
Researchers receive over $40,000 to study role of personality and resiliency in Veteran mental health
LONDON, ON – The unique and challenging experiences faced by Canadian Armed Forces (CAF) Veterans puts this group at high risk for developing symptoms of mental health disorders, such as Post traumatic stress disorder (PTSD), depression, and anxiety. Researchers at Lawson Health Research Institute (Lawson) have been awarded over $40,000 from Veterans Affairs Canada to study various risk and protective factors associated with mental health disorders in CAF Veterans.
The study aims to learn whether an individual’s personality traits serve as risk or protective factors for Veterans developing mental health problems. The traits that will be examined include openness, agreeableness, emotionality, honesty-humility, conscientiousness, extraversion, and resiliency. For example, someone who is high in openness and resiliency may have a lower risk of developing mental health symptoms.
“Veterans and military members experience uniquely challenging events when compared to the general population,” explains Dr. Rachel Plouffe, study co-lead and Postdoctoral Associate at Lawson. “Past research has identified some risk factors, including history of childhood trauma, age, gender, and education. However, no one to date has investigated the role that personality traits might play in Veterans’ symptoms of mental health conditions.”
The research team will look at the relationship between these personality traits, exposure to stressful experiences during military operations, and mental health outcomes through survey data provided by 500 treatment-seeking and non-treatment seeking Veterans. This information will be used to assess whether the individual’s expression of these personality traits can predict levels of depression, anxiety and PTSD.
They also hope to determine whether the association between combat exposure and PTSD is stronger for those higher in traits such as emotionality, which reflects those who tend to be anxious and fearful. On the other hand, this association will likely be weaker for those higher in more “positive” traits, such as agreeableness, as these individuals tend to be cooperative and forgiving toward others.
Plouffe notes that personalities are not completely stagnant. Certain traits can often change over time, and individuals can work to increase their levels of resiliency.
“Ultimately, our hope is to help improve the ability of CAF members and Veterans to thrive even in the face of great adversity. With the findings from our research, military organizations and mental health treatment centres could use the information from this study to predict which patients may be at greater risk of developing mental health disorders, and provide tailored treatment for these individuals,” she explains.
“This research is needed for military organizations to improve and implement strategies that enhance the well-being of Veterans and active service members,” says Dr. Don Richardson, study co-lead and Director of the MacDonald Franklin OSI Research Centre at Lawson. “Our Veterans give a lot to serve and protect, so it is our duty as researchers and clinicians to find ways that we can better serve them.”
-30-
Lawson Health Research Institute is one of Canada’s top hospital-based research institutes, tackling the most pressing challenges in health care. As the research institute of London Health Sciences Centre and St. Joseph’s Health Care London, our innovation happens where care is delivered. Lawson research teams are at the leading-edge of science with the goal of improving health and the delivery of care for patients. Working in partnership with Western University, our researchers are encouraged to pursue their curiosity, collaborate often and share their discoveries widely. Research conducted through Lawson makes a difference in the lives of patients, families and communities around the world. To learn more, visit www.lawsonresearch.ca.
Senior Media Relations Consultant
Communications & Public Engagement
T: 519-685-8500 ext. 73502
Celine.zadorsky@lhsc.on.ca
Researchers receive over $40,000 to study role of personality and resiliency in Veteran mental health
The unique and challenging experiences faced by Canadian Armed Forces (CAF) Veterans puts this group at high risk for developing symptoms of mental health disorders, such as Post traumatic stress disorder (PTSD), depression, and anxiety. Researchers at Lawson Health Research Institute (Lawson) have been awarded over $40,000 from Veterans Affairs Canada to study various risk and protective factors associated with mental health disorders in CAF Veterans.
The study aims to learn whether an individual’s personality traits serve as risk or protective factors for Veterans developing mental health problems. The traits that will be examined include openness, agreeableness, emotionality, honesty-humility, conscientiousness, extraversion, and resiliency. For example, someone who is high in openness and resiliency may have a lower risk of developing mental health symptoms.
“Veterans and military members experience uniquely challenging events when compared to the general population,” explains Dr. Rachel Plouffe, study co-lead and Postdoctoral Associate at Lawson. “Past research has identified some risk factors, including history of childhood trauma, age, gender, and education. However, no one to date has investigated the role that personality traits might play in Veterans’ symptoms of mental health conditions.”
The research team will look at the relationship between these personality traits, exposure to stressful experiences during military operations, and mental health outcomes through survey data provided by 500 treatment-seeking and non-treatment seeking Veterans. This information will be used to assess whether the individual’s expression of these personality traits can predict levels of depression, anxiety and PTSD.
They also hope to determine whether the association between combat exposure and PTSD is stronger for those higher in traits such as emotionality, which reflects those who tend to be anxious and fearful. On the other hand, this association will likely be weaker for those higher in more “positive” traits, such as agreeableness, as these individuals tend to be cooperative and forgiving toward others.
Plouffe notes that personalities are not completely stagnant. Certain traits can often change over time, and individuals can work to increase their levels of resiliency.
“Ultimately, our hope is to help improve the ability of CAF members and Veterans to thrive even in the face of great adversity. With the findings from our research, military organizations and mental health treatment centres could use the information from this study to predict which patients may be at greater risk of developing mental health disorders, and provide tailored treatment for these individuals,” she explains.
“This research is needed for military organizations to improve and implement strategies that enhance the well-being of Veterans and active service members,” says Dr. Don Richardson, study co-lead and Director of the MacDonald Franklin OSI Research Centre at Lawson. “Our Veterans give a lot to serve and protect, so it is our duty as researchers and clinicians to find ways that we can better serve them.”
Researchers seek Canadian health care workers for study on moral distress during COVID-19 pandemic
LONDON, ON - A team from Lawson Health Research Institute is seeking 500 Canadian health care workers to participate in a study on moral distress and psychological wellbeing during the COVID-19 pandemic. Participants will complete online surveys once every three months for a total of 18 months. The goal is to better understand the pandemic’s impact on health care workers in order to minimize moral distress and support wellbeing during future pandemic events.
Moral distress is a form of psychological distress that occurs following an event that conflicts with a person’s moral values or standards. Through previous research with military populations, moral distress has been linked to an increased risk of post-traumatic stress disorder (PTSD) and depression.
“Health care workers are facing unprecedented demands as a result of the pandemic and many may be working under extreme physical and psychological stress,” says Dr. Don Richardson, Lawson Associate Scientist and Director of the MacDonald Franklin Operational Stress Injury (OSI) Research Centre. “Health care workers may face difficult moral-ethical decisions including those around patient care and shortages of personal protective equipment (PPE), which could lead to moral distress.”
The study will consist of a series of surveys to assess moral distress during the pandemic. Participating health care workers will answer questions about moral-ethical dilemmas and symptoms of depression, PTSD, general anxiety, and burnout.
“Moral-ethical dilemmas must be considered in the context of other difficulties faced by health care workers during the pandemic, such as increased workloads, reduced social activities, and evolving work environments and health care delivery models,” explains Dr. Anthony Nazarov, Associate Scientist at Lawson and the MacDonald Franklin OSI Research Centre.
The team hopes that results can be used to cultivate wellness at the outset of future pandemics. This might include guiding emergency preparedness policies and moral-ethical decision-making training modules. They hope that by tracking psychological outcomes over time, they can identify early warning signs of distress that can be targeted with early interventions.
The researchers will also ask questions that explore how the pandemic is impacting health care delivery, such as increased reliance on virtual care appointments, and whether health care workers are satisfied with these changes.
“It is necessary to provide a voice to health care workers during this pandemic,” adds Dr. Richardson. “This is the first study to measure the moral-ethical dilemmas faced in a pandemic and the impacts of such dilemmas on moral distress. It is also the first to measure health care worker perceptions on the virtual migration of patient care.”
This project is in partnership with the Centre of Excellence on PTSD and Related Mental Health Conditions. The focus of the Centre of Excellence is to create knowledge networks across Canada to increase expertise and create the best possible supports and services for Veterans, first responders and their families.
Interested Canadian health care workers can learn more about the study and access the survey at https://participaid.co/studies/bYE4Ob.
-30-
Lawson Health Research Institute is one of Canada’s top hospital-based research institutes, tackling the most pressing challenges in health care. As the research institute of London Health Sciences Centre and St. Joseph’s Health Care London, our innovation happens where care is delivered. Lawson research teams are at the leading-edge of science with the goal of improving health and the delivery of care for patients. Working in partnership with Western University, our researchers are encouraged to pursue their curiosity, collaborate often and share their discoveries widely. Research conducted through Lawson makes a difference in the lives of patients, families and communities around the world. To learn more, visit www.lawsonresearch.ca.
Senior Media Relations Consultant
Communications & Public Engagement
T: 519-685-8500 ext. 73502
Celine.zadorsky@lhsc.on.ca
Researchers seek Canadian health care workers for study on moral distress during COVID-19 pandemic
Researchers from Lawson Health Research Institute and the Centre of Excellence on PTSD and Related Mental Health Conditions are seeking 500 Canadian health care workers to participate in a study on moral distress and psychological wellbeing during the COVID-19 pandemic. Participants will complete online surveys once every three months for a total of 18 months. The goal is to better understand the pandemic’s impact on health care workers in order to minimize moral distress and support wellbeing during future pandemic events.
Moral distress is a form of psychological distress that occurs following an event that conflicts with a person’s moral values or standards. Through previous research with military populations, moral distress has been linked to an increased risk of post-traumatic stress disorder (PTSD) and depression.
“Health care workers are facing unprecedented demands as a result of the pandemic and many may be working under extreme physical and psychological stress,” says Dr. Don Richardson, Lawson Associate Scientist and Director of the MacDonald Franklin Operational Stress Injury (OSI) Research Centre. “Health care workers may face difficult moral-ethical decisions including those around patient care and shortages of personal protective equipment (PPE), which could lead to moral distress.”
The study will consist of a series of surveys to assess moral distress during the pandemic. Participating health care workers will answer questions about moral-ethical dilemmas and symptoms of depression, PTSD, general anxiety, and burnout.
“Moral-ethical dilemmas must be considered in the context of other difficulties faced by health care workers during the pandemic, such as increased workloads, reduced social activities, and evolving work environments and health care delivery models,” explains Dr. Anthony Nazarov, Associate Scientist at Lawson and the MacDonald Franklin OSI Research Centre.
The team hopes that results can be used to cultivate wellness at the outset of future pandemics. This might include guiding emergency preparedness policies and moral-ethical decision-making training modules. They hope that by tracking psychological outcomes over time, they can identify early warning signs of distress that can be targeted with early interventions.
The researchers will also ask questions that explore how the pandemic is impacting health care delivery, such as increased reliance on virtual care appointments, and whether health care workers are satisfied with these changes.
“It is necessary to provide a voice to health care workers during this pandemic,” adds Dr. Richardson. “This is the first study to measure the moral-ethical dilemmas faced in a pandemic and the impacts of such dilemmas on moral distress. It is also the first to measure health care worker perceptions on the virtual migration of patient care.”
Researchers testing triple intervention to combat dementia
Researchers at Lawson Health Research Institute are the first in the world conducting a clinical trial to test a triple intervention aimed at treating Mild Cognitive Impairment (MCI) and delaying the onset of dementia. The Mobility, Exercise and Cognition (MEC) team will be incorporating physical exercises, cognitive training and vitamin D supplementation to determine the best treatment for improving mobility and cognition.
“We have learned the brain processes involved in motor-control - for example how a person walks - and cognition - for example how that person solves a problem - share similar locations and networks in the brain,” explains Dr. Manuel Montero Odasso, Lawson Scientist and Geriatrician at St. Joseph’s Health Care London. “Problems with mobility are connected to lowering function in the mind, and so can be a good indicator of future progression into dementia.”
Dr. Montero Odasso is also an Associate Professor in the departments of Medicine and Epidemiology and Biostatistics at the Schulich School of Medicine & Dentistry, Western University.

Gait assessment looks at the way in which we move our whole body from one point to another, helping to analyze mobility and the brain processes involved.
MCI is an intermediate stage between the expected cognitive decline of normal aging and the more serious decline of dementia. It can involve problems with memory, language, thinking and judgment. While many older individuals experience decline in both mobility and cognition, each are assessed and treated separately with no specific recommendations available for physicians.
The SYNERGIC Trial will combine physical exercises, cognitive training and vitamin D to test how these interventions work together to improve cognition in older adults at risk for dementia. The trial is targeting cognitive decline at the earliest stage, individuals with MCI, where interventions are more likely to have an effect and can be monitored.

Dr. Manuel Montero Odasso, Lawson Scientist and lead for the SYNERGIC Trial.
Dr. Montero Odasso explains that both physical and cognitive exercises have shown promising effects for maintaining cognition, while vitamin D deficiency is associated with cognitive decline. A key feature of this trial is that participants will receive individualized and progressive training.
“By delaying declines in cognition, we can improve a person’s quality of life. This research will help to support a more comprehensive preventative treatment with clinical guidelines for physicians whose patients are at risk of developing dementia,” states Dr. Montero Odasso. “Even more, each one year delay of progression to dementia in older individuals at risk has the opportunity to save billions of dollars for the Canadian health care system.”
Individuals over 60 years old with mild cognitive impairment without dementia are eligible for this clinical trial. Those interested in participating are encouraged to contact Research Coordinator Alanna Black at 519.685.4292 ext. 42179.
Participants will be asked to complete a routine of exercises and cognitive training three times a week for six months, with one final assessment at 12 months. The main site for the study is Parkwood Institute with physical exercises taking place at the Labatt Health Sciences Building at Western University, in Dr. Kevin Shoemaker’s Laboratory for Brain and Heart Health.
This study has been funded by the Canadian Consortium on Neurodegenerative in Aging (CCNA) which represents Canada-wide research aimed at enhancing the quality of life and services for individuals diagnosed with a neurodegenerative disease. The MEC team in London, led by Dr. Montero Odasso, includes expert researchers in the field of mobility who aim to develop common assessments for the interaction of cognition and mobility for older people to aid as a diagnostic tool for detecting dementia.

Members of the study’s research team, from left to right: Korbin Blue, Research Assistant (Co-op Student); Yanina Sarquis-Adamson, Lab Research Assistant; Frederico Faria, Post-Doctoral Fellow; Dr. Montero Odasso, Director, Gait and Brain Lab; research participant; Alanna Black, Lab Research Coordinator; Stephanie Cullen, Research Assistant (Undergraduate Student); and, Navena Lingum, Research Assistant (Master Student).
Researchers to study inhaled sedatives as solution to COVID-19 drug shortages
A team from Lawson Health Research Institute are being funded by the Government of Ontario’s COVID-19 Rapid Research Fund to study whether inhaled sedatives can replace those that are delivered intravenously in COVID-19 patients requiring ventilation. The multi-centre clinical trial aims to address a global shortage of intravenous (IV) sedatives while improving patient outcomes.
“When COVID-19 patients develop severe respiratory failure and need to be ventilated, they require sedation. While IV sedatives are currently used, there is concern about global drug shortages, particularly if there’s a second wave of COVID-19 in the fall,” explains Dr. Marat Slessarev, Scientist at Lawson and Critical Care Physician at London Health Sciences Centre (LHSC). “Even if we have enough ventilators, we won’t be able to ventilate patients without sedatives.”
The clinical trial, being co-led by Dr. Slessarev and Dr. Angela Jerath at Sunnybrook Health Sciences Centre, will study the replacement of IV sedatives with inhaled sedatives. Inhaled sedatives, also called volatiles, are widely available due to their use in operating rooms to sedate patients during surgery. While they have not been routinely used to sedate patients needing ventilation, early studies suggest they could be safe and even more effective than IV sedatives.
“Preliminary studies in non-COVID patients with severe respiratory failure suggest that inhaled sedatives can reduce lung inflammation, shorten the duration of ventilation and potentially improve survival. Inhaled sedatives could therefore reduce the pandemic’s strain on ventilator capacity while improving patient outcomes,” says Dr. Slessarev. “Since these drugs are safe, cheap and readily available, they can easily be used to address IV sedative shortages if found effective.”
The researchers will recruit approximately 800 patients from across Canada and the United States including patients from LHSC. Each patient will be randomized to receive either IV sedatives or inhaled sedatives. Patient outcomes such as survival and length of ventilation will be compared between the two groups to determine which method of sedation is most effective.
“This is the largest trial of its kind. If inhaled sedatives can shorten the length of ventilation or improve survival in patients with serve respiratory failure, this could cause a paradigm shift in the way we sedate patients in intensive care units (ICUs) around the world,” notes Dr. Slessarev.
Given many survivors of critical illness experience cognitive impairment for months or even years after an intensive care unit (ICU) stay, the team is also planning a sub-study to assess whether one method of sedation results in better cognitive outcomes after treatment.
In addition to funding from the Government of Ontario, the study is being supported with funding from the Canadian Institutes of Health Research (CIHR), London Health Sciences Foundation and Sunnybrook Health Sciences Centre.

Dr. Marat Slessarev, Scientist at Lawson and Critical Care Physician at LHSC
Researchers to study inhaled sedatives as solution to COVID-19 drug shortages
LONDON, ON - A team from Lawson Health Research Institute are being funded by the Government of Ontario’s COVID-19 Rapid Research Fund to study whether inhaled sedatives can replace those that are delivered intravenously in COVID-19 patients requiring ventilation. The multi-centre clinical trial aims to address a global shortage of intravenous (IV) sedatives while improving patient outcomes.
“When COVID-19 patients develop severe respiratory failure and need to be ventilated, they require sedation. While IV sedatives are currently used, there is concern about global drug shortages, particularly if there’s a second wave of COVID-19 in the fall,” explains Dr. Marat Slessarev, Scientist at Lawson and Critical Care Physician at London Health Sciences Centre (LHSC). “Even if we have enough ventilators, we won’t be able to ventilate patients without sedatives.”
The clinical trial, being co-led by Dr. Slessarev and Dr. Angela Jerath at Sunnybrook Health Sciences Centre, will study the replacement of IV sedatives with inhaled sedatives. Inhaled sedatives, also called volatiles, are widely available due to their use in operating rooms to sedate patients during surgery. While they have not been routinely used to sedate patients needing ventilation, early studies suggest they could be safe and even more effective than IV sedatives.
“Preliminary studies in non-COVID patients with severe respiratory failure suggest that inhaled sedatives can reduce lung inflammation, shorten the duration of ventilation and potentially improve survival. Inhaled sedatives could therefore reduce the pandemic’s strain on ventilator capacity while improving patient outcomes,” says Dr. Slessarev. “Since these drugs are safe, cheap and readily available, they can easily be used to address IV sedative shortages if found effective.”
The researchers will recruit approximately 800 patients from across Canada and the United States including patients from LHSC. Each patient will be randomized to receive either IV sedatives or inhaled sedatives. Patient outcomes such as survival and length of ventilation will be compared between the two groups to determine which method of sedation is most effective.
“This is the largest trial of its kind. If inhaled sedatives can shorten the length of ventilation or improve survival in patients with serve respiratory failure, this could cause a paradigm shift in the way we sedate patients in intensive care units (ICUs) around the world,” notes Dr. Slessarev.
Given many survivors of critical illness experience cognitive impairment for months or even years after an intensive care unit (ICU) stay, the team is also planning a sub-study to assess whether one method of sedation results in better cognitive outcomes after treatment.
In addition to funding from the Government of Ontario, the study is being supported with funding from the Canadian Institutes of Health Research (CIHR), London Health Sciences Foundation and Sunnybrook Health Sciences Centre.
-30-
DOWNLOADABLE MEDIA

Dr. Marat Slessarev, Scientist at Lawson Health Research Institute and Critical Care Physician at London Health Sciences Centre (LHSC)
Lawson Health Research Institute is one of Canada’s top hospital-based research institutes, tackling the most pressing challenges in health care. As the research institute of London Health Sciences Centre and St. Joseph’s Health Care London, our innovation happens where care is delivered. Lawson research teams are at the leading-edge of science with the goal of improving health and the delivery of care for patients. Working in partnership with Western University, our researchers are encouraged to pursue their curiosity, collaborate often and share their discoveries widely. Research conducted through Lawson makes a difference in the lives of patients, families and communities around the world. To learn more, visit www.lawsonresearch.ca.
Senior Media Relations Consultant
Communications & Public Engagement
T: 519-685-8500 ext. 73502
Celine.zadorsky@lhsc.on.ca
Researchers unravel two mysteries of COVID-19
LONDON, ONTARIO - A team from Lawson Health Research Institute and Western University has made significant steps forward in understanding COVID-19 through two back-to-back studies published this week in Critical Care Explorations. In one study, the team has identified six molecules that can be used as biomarkers to predict how severely ill a patient will become. In the other study, they are the first to reveal a new mechanism causing blood clots in COVID-19 patients and potential ways to treat them.
The studies were conducted by analyzing blood samples from critically ill patients at London Health Sciences Centre (LHSC). They build on a growing body of work from the team who were first in the world to profile the body’s immune response to the virus by revealing a separate six molecules that could act as potential targets to treat hyperinflammation in critically ill patients.
“We’ve begun answering some of the biggest COVID-19 questions asked by clinicians and health researchers,” says Dr. Douglas Fraser, lead researcher from Lawson and Western’s Schulich School of Medicine & Dentistry, and Critical Care Physician at LHSC. “While the findings need to be validated with larger groups of patients, they could have important implications for treating and studying this disease.”
Predicting which COVID-19 patients will get worse
With no proven therapies, many COVID-19 patients admitted to intensive care units (ICUs) do not survive.
“When a patient is admitted to ICU, we normally wait to see if they are going to get worse before we consider any risky interventions. To improve outcomes, we not only need new therapies but also a way to predict prognosis or which patients are going to get worse,” explains Dr. Fraser.
The researchers identified six molecules of importance (CLM-1, IL12RB1, CD83, FAM3B, IGFR1R and OPTC). They found that these molecules were elevated in COVID-19 patients who would become even more severely ill. They found that when measured on a COVID-19 patient’s first day of ICU admission, the molecules could be used to predict which patients will survive following standard ICU treatment.
“While further research is needed, we’re confident in these biomarkers and suspect these patterns may be present even before ICU admission, such as when a patient first presents to the emergency department,” notes Dr. Fraser. “These findings could be incredibly important in determining how severely ill a patient will become.”
The team measured 1,161 plasma proteins from the blood of 30 participants: 10 COVID-19 patients and 10 patients with other infections admitted to LHSC’s ICU, as well as 10 healthy control participants. Blood was drawn on set days of ICU admission, processed in a lab and then analyzed using statistical methods and artificial intelligence.
The team notes that predicting a patient’s disease severity can help in a number of ways. It could allow for medical teams to have important conversations with family members, setting goals of care based on the patient’s health and personal wishes. Medical teams could use the knowledge to mobilize resources more quickly. If they know a patient is at higher risk of death, they may consider intervening sooner despite associated risks. The team also hopes the findings can be used to better design COVID-19 clinical trials by grouping patients based on their risk. This could allow for stronger results when examining potential treatments for the disease.
Understanding why blood clots occur and how to treat them
A major complication occurring in most critically ill COVID-19 patients is clotting in the lung’s small blood vessels which leads to low oxygen levels in the body.
“The reason for this clotting has been unclear. Most suspect the clotting mechanisms in our blood are put into overdrive and so many clinicians have been treating with anticoagulant therapies like the drug heparin,” says Dr. Fraser. “But we’ve uncovered an entirely different mechanism.”
The team further analyzed the blood samples from their 30 participants, and found evidence to suggest that the inner linings of small blood vessels are becoming damaged and inflamed, making them a welcoming environment for platelets (small blood cells) to stick.
They discovered that COVID-19 patients had elevated levels of three molecules (hyaluronic acid, syndecan-1 and P-selectin.) The first two molecules are products broken down from small hair-like structures (the glycocalyx) which line the inside of the blood vessels. Their presence suggests the glycocalyx is being damaged with its breakdown products sent into the bloodstream. The presence of P-selectin is also significant as this molecule helps to make both platelets and the inner lining of blood vessels adhere to one another.
“The glycocalyx keeps platelets from touching the inside wall of the blood vessel and helps facilitate the production of nitric oxide, which has an important role in preventing platelets from sticking,” explains Dr. Fraser. “We suspect the body’s immune response is producing enzymes that shear off these little hair-like structures, inflaming blood vessels and making them a welcoming environment for platelets to form clots.”
The team suggests that two therapies may hold promise for treating blood clots in COVID-19 patients: platelet inhibitors to stop platelets from sticking and molecules to protect and restore the inner lining of blood vessels.
“By exploring these therapies as potential alternatives to anticoagulant therapies, we may be able to improve patient outcomes,” says Dr. Fraser. “Through our combined findings, we hope to provide tools to predict which patients will become the most severely ill and treatments for both hyperinflammation and blood clots.”
-30-
DOWNLOADABLE MEDIA


Dr. Douglas Fraser, Researcher from Lawson Health Research Institute and Western University’s Schulich School of Medicine & Dentistry
Lawson Health Research Institute is one of Canada’s top hospital-based research institutes, tackling the most pressing challenges in health care. As the research institute of London Health Sciences Centre and St. Joseph’s Health Care London, our innovation happens where care is delivered. Lawson research teams are at the leading-edge of science with the goal of improving health and the delivery of care for patients. Working in partnership with Western University, our researchers are encouraged to pursue their curiosity, collaborate often and share their discoveries widely. Research conducted through Lawson makes a difference in the lives of patients, families and communities around the world. To learn more, visit www.lawsonresearch.ca.
The Schulich School of Medicine & Dentistry at Western University is one of Canada’s preeminent medical and dental schools. Established in 1881, it was one of the founding schools of Western University and is known for being the birthplace of family medicine in Canada. For more than 130 years, the School has demonstrated a commitment to academic excellence and a passion for scientific discovery.
Senior Media Relations Consultant
Communications & Public Engagement
T: 519-685-8500 ext. 73502
Celine.zadorsky@lhsc.on.ca
Researching treatments for COVID-19
As we continue to live with a COVID-19 pandemic, patients will need good treatment options.
Hospital researchers in London, Ontario, through Lawson Health Research Institute, are testing treatment options for patients who have been hospitalized from a COVID-19 infection, often for severe symptoms.
During the peaks of the COVID-19 pandemic, there were concerns of a global drug shortage when it came to IV sedatives for patients needing ventilation. Through funding from the Government of Ontario’s COVID-19 Rapid Research Fund, a team of Ontario researchers studied whether inhaled sedatives could replace those that are delivered through IV.
“In addition to easing the burden on IV stocks, the inhaled sedatives have additional benefits,” says Dr. Marat Slessarev, a Critical Care Physician at LHSC. “They may reduce inflammation in the lungs and shorten the duration of sedation because they are eliminated from the body faster than IV sedatives.”
Dr. Slessarev, also a Scientist at Lawson, adds that using inhaled sedatives could also be safer for severe COVID-19 patients, who in many cases are on a ventilator for a long time. “When using IV sedatives, they can accumulate and metabolize with accumulation. This can leave the patient with the potential of developing kidney issues because it is hard to dispel them quickly.”

Dr. Marat Slessarev, Critical Care Physican at LHSC and Lawson Scientist
To date, around 800 patients across ten hospital sites have been enrolled in this trial, with the hopes of getting more hospital sites on board as the clinical trials continue.
A majority of people with severe COVID-19 infections in critical care end up developing sepsis, which is a potentially life-threatening condition that occurs when the body's response to an infection damages its own tissues.
“An inflammatory response is what the body uses to fight an infection, but sometimes it is more than necessary and it causes damage to the organs and the body,” explains Dr. Claudio Martin, a Physician in the Intensive Care Unit (ICU) at London Health Sciences Centre (LHSC). “This is why a patient with a severe COVID-19 infection caused by a virus can get sepsis.”
Dr. Martin, who is also an Associate Scientist at Lawson, began studying the use of a human protein called Annexin A5 as a potential treatment for COVID-19 patients with sepsis. “It can work in two ways. The annexin may coat injured cells and reduce the inflammatory response. Or, the injured cells and exposed proteins trigger the clotting mechanism in the body. Those with COVID-19 could get blood clots in the brain and the lungs, and using annexin may improve their outcomes.”

Dr. Claudio Martin, Critical Care Physician at LHSC and Lawson Scientist
Currently, a clinical trial using Annexin A5 on severe COVID-19 patients is underway at LHSC to look at the efficacy of using this human protein.
The COVID-19 virus is also known to cause respiratory failure, prompting Dr. Jim Lewis, Respirologist at St. Joseph’s Health Care London and Scientist at Lawson, to investigate the use of pulmonary surfactant as a potential treatment for these patients.
Bovine Lipid Extract Surfactant Suspension (BLES) is a pulmonary surfactant manufactured in London, Ontario. It’s currently used worldwide to help improve lung function in premature babies. Now, it is being studied with COVID-19 patients experiencing respiratory failure.

Dr. Jim Lewis, Repirologist at St. Joseph's Health Care London and Lawson Scientist
“We know that patients who have injury to their lung and require ventilation have inflammation in the airways of their lungs. Surfactant is a homogeneous layer of lipids and proteins that line the lungs to allow us to breath with minimal effort,” explains Dr. Lewis. “If we gave these patients surfactant as soon as possible after they were put on a mechanical ventilator, it may have some benefit in improving their outcome and getting them off the ventilator sooner.”
Dr. Lewis and his team conducted a clinical trial using pulmonary surfactant with ten critically ill COVID-19 patients and initial results have shown patient safety and efficacy.
Another team of hospital researchers turned to using modified dialysis machines to offer treatment options to those with severe COVID-19 symptoms. Nephrologist at LHSC, Dr. Chris McIntyre, was the first in the world to modify a dialysis device to treat a patient with COVID-19. The device gently removes a patient’s blood, modifies white blood cells, and returns them to fight hyperinflammation.
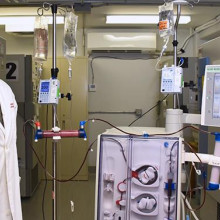
Dr. Chris McIntyre, Nephrologist at LHSC and Lawson Scientist
“It was quick. We went from the initial idea to the approvals, creating the device and appointing the first patient within 40 days,” says Dr. McIntyre who is also a Scientist at Lawson. “After the first 12 patients, we found that those treated with the device needed significantly less drugs to maintain their blood pressure. We were able to deliver the treatment for all the patients – none of the treatments failed and we had no safety issues.”
Dr. McIntyre is now looking into using this form of therapy for chronic dialysis patients to help modify organ injury.