Search
Search
Assessing the pandemic's impact on Canadian Veterans and their spouses
Multiple studies are reporting the pandemic’s impact on the mental health of Canadians, but what effect is it having on our nation’s Veterans and their spouses?
“With concerns about COVID-19 infection and drastic changes to everyday life, the pandemic is taking a toll on the health of Canadians,” explains Dr. Don Richardson, Associate Scientist at Lawson Health Research Institute and Director of the MacDonald Franklin Operational Stress Injury (OSI) Research Centre, located at St. Joseph's Health Care London's Parkwood Institute. “And it may be particularly distressing for those vulnerable to mental illness.”
Population studies show that Veterans are at double the risk of mental illness when compared to the rest of the population. They experience higher rates of depression, anxiety and loneliness. Spouses of Canadian Veterans are also at higher risk of distress, as they may sometimes undertake significant caregiving responsibilities that that require significant emotional and time investments.
“It’s currently unknown how the pandemic will impact Veterans and their spouses, but it could result in particularly serious outcomes,” says Dr. Anthony Nazarov, Associate Scientist at Lawson and the MacDonald Franklin OSI Research Centre.
In a new project from Lawson and the Centre of Excellence on Post-Traumatic Stress Disorder (PTSD), researchers will seek answers to these questions by partnering with up to 1,000 Canadian Veterans and 250 spouses of Canadian Veterans. Through online surveys, the project will hear directly from Veterans and their spouses to assess the pandemic’s effects on their well-being over time. The team hopes results can be used by health care workers and policy-makers to support Veterans and their families during both the current pandemic and future public health emergencies.
“We want to hear from all Canadian Veterans and their spouses, whether they’re doing well or not and whether they’re seeking care or not,” explains Dr. Nazarov.

From left: Drs. Don Richardson and Anthony Nazarov
Participants will complete online surveys, available in both English and French, once every three months for a total of 18 months. They will be asked questions about their psychological, social, family-related and physical well-being, and any relevant changes to their lifestyle and health care treatment.
“Veterans who regularly access health care services could encounter significant changes, including a move to virtual care appointments. This could lead to increased caregiving responsibilities for spouses,” says Dr. Nazarov. “Given the uncertainty surrounding the pandemic, these changes may persist well into the future, mandating a thorough assessment of patient satisfaction and treatment outcomes.”
The team hopes results can be used to support the wellness of Veterans and their families during public health emergencies. This includes providing health care professionals and policy-makers with information to guide emergency preparedness policies and health care delivery models. They hope results can also be used to recognize early signs of distress in order to target with early interventions.
“We are seeking to understand the impact of COVID-19 on Veterans and their families to identify if this global pandemic is leading to psychological distress or triggering historical traumas,” says Dr. Patrick Smith, CEO of the Centre of Excellence on PTSD. “The Centre’s primary goal is to increase Canadian expertise related to military and Veteran mental health, suicide prevention and substance use disorders. This study can help us understand if the pandemic is having debilitating and life-altering effects, and help us address a potential mental health crisis.”

Dr. Patrick Smith
Benchmarking the evaluation process for living kidney donation to improve outcomes
LONDON, ONTARIO - Living kidney donation rates have remained stagnant over the last decade, and many potential living donors find the evaluation process to be lengthy and difficult to navigate. Researchers from Lawson Health Research Institute and Western University have published results identifying indicators to monitor and improve the performance of transplant centres in how efficiently they evaluate individuals interested in becoming living kidney donors. The findings appear in the current issue of CJASN.
In 2019, there were 219 living donor kidney transplants in Ontario. There are several advantages to kidney transplantation from a living donor compared to deceased. However, many living donor candidates drop out of the evaluation process for a variety of reasons.
“From speaking with living donors, we know that some people find the evaluation process frustrating and cumbersome,” says Dr. Amit Garg, Professor at Western’s Schulich School of Medicine & Dentistry and Scientist at Lawson. “To ensure living donation is safe for both the donor and the recipient, a thorough evaluation is needed looking at medical, physical and psychosocial considerations.” Dr. Garg is also Director of the Living Kidney Donor Program at London Health Sciences Centre (LHSC).
Marian Reich started the journey of living kidney donation 11 years ago, giving the gift of life to her sister-in-law, Susan. “The evaluation process took one full year to complete, and in that time Susan lost the option of a pre-emptive transplant and started dialysis. I wasn’t fully aware that I had to be the driver of the process, and how to do that.”
Reich believes living donor evaluation should be consistent across the country, with a clearer process outlined for potential living donors and recipients. “Research studies like this one bring forth best practice and evidence-based information that includes the perspective of those with lived experiences.”
The research team, which included Patient Partners such as Reich who co-authored the paper, surveyed 77 participants who were kidney transplant recipients and recipient candidates, living kidney donors and donor candidates, or health care providers and administrators.
Out-of-pocket cost incurred by donors is one of the most significant barriers to donation. Another challenge is the lack of reliable information about the expectations and requirements of the donation process. For example, the time commitment, nature of the testing and reasons for the tests performed.
The evaluation process takes on average 10 months to complete, requiring multiple visits to the transplant centre. The intended recipient’s health may deteriorate during this wait to the point they may no longer receive a transplant. “By improving the efficiency of the process, more living donor candidates could complete the evaluation and more transplants performed,” adds Dr. Garg.
The study found a diverse set of quality indicators to measure the evaluation process, focusing on efficiency and safety to improve patient outcomes. They also identified a single measure tracking the costs of living donor evaluation from a health system perspective to assist with planning and budgeting.
“This is an important first step towards system monitoring, benchmarking and accountability in living kidney donor evaluation,” explains Dr. Steven Habbous, co-lead investigator and PhD graduate with Lawson and Western’s Schulich School of Medicine & Dentistry. “In other areas of the health care system, there are reported metrics that hospitals and funders use to compare performance between similar hospitals and track changes over time. We need to measure what we value, and value what we measure.”
The team will present the data to the Canadian National Living Kidney Donation Advisory Committee to help develop national guidelines. Potential improvement strategies are to evaluate multiple living donor candidates concurrently for the same recipient or offer some aspects of the evaluation virtually.
The study was funded by the Can-SOLVE CKD Network, a Canada-wide initiative to enhance kidney disease research and care in partnership with patients. The Ontario-based team led by Dr. Garg is one of 18 Can-SOLVE CKD research groups across the country investigating issues such as earlier diagnosis, better treatments, and innovative care – all based on priorities identified by patients.
-30-
Downloadable Media
Click image for larger version

Dr. Amit Garg, study co-lead investigator, Professor at Western’s Schulich School of Medicine & Dentistry, and Scientist at Lawson. Dr. Garg is also Director of the Living Kidney Donor Program at LHSC.

Dr. Steven Habbous, study co-lead investigator and PhD graduate with Lawson and Western’s Schulich School of Medicine & Dentistry

Marian Reich, study co-author, Patient Partner and living kidney donor
Lawson Health Research Institute is one of Canada’s top hospital-based research institutes, tackling the most pressing challenges in health care. As the research institute of London Health Sciences Centre and St. Joseph’s Health Care London, our innovation happens where care is delivered. Lawson research teams are at the leading-edge of science with the goal of improving health and the delivery of care for patients. Working in partnership with Western University, our researchers are encouraged to pursue their curiosity, collaborate often and share their discoveries widely. Research conducted through Lawson makes a difference in the lives of patients, families and communities around the world. To learn more, visit www.lawsonresearch.ca.
The Schulich School of Medicine & Dentistry at Western University is one of Canada’s preeminent medical and dental schools. Established in 1881, it was one of the founding schools of Western University and is known for being the birthplace of family medicine in Canada. For more than 130 years, the School has demonstrated a commitment to academic excellence and a passion for scientific discovery.
Senior Media Relations Consultant
Communications & Public Engagement
T: 519-685-8500 ext. 73502
Celine.zadorsky@lhsc.on.ca
Beneficial bacteria may protect against breast cancer
Dr. Gregor Reid, a scientist at Lawson Health Research Institute, and his Western University PhD student, Camilla Urbaniak, have previously shown that live bacteria are present in the breast tissues of healthy women. This proves the existence of a breast tissue microbiome. In past studies, Reid and Urbaniak have also proven that human milk contains beneficial bacteria. “Since breastfeeding decreases a woman’s risk for breast cancer, we wondered if beneficial bacteria, like those found in human milk, may be playing a role in lowering the risk of cancer and whether other types of bacteria could be influencing cancer formation,” said Dr. Reid.
To explore these questions, Urbaniak obtained breast tissue samples from 58 women who had either benign or cancerous tumours. In addition, she obtained 23 samples from healthy women undergoing breast reductions or enhancements. Through an analysis of these tissues, Urbaniak found that the bacteria present in the breasts of healthy women differ from those found in the breasts of women with breast cancer.
Women with breast cancer had elevated levels of both Escherichia coli (E. coli) and Staphylococcus epidermidis. A NASA study has confirmed the London research findings and further identified bacteria associated with breast cancer. Urbaniak and Reid went even further by showing that these bacteria can cause significant damage, known as double-stranded breaks, to DNA. When this occurs, the body tries to repair the damage. However, these repairs often result in errors which can lead to the development of cancer.
Tissues taken from the breasts of healthy women showed high levels of Lactobacillus and Streptococcus, known to promote health and display characteristics that can prevent cancer. For example, Streptococcus produces antioxidants that can help prevent DNA damage.
“This study provides clear evidence that the breast tissue microbiome differs between healthy women and those with breast cancer,” said Dr. Reid. “Our colleagues in Spain have recently shown that probiotics with lactobacilli can be ingested by women and reach the mammary gland. This raises the questions of whether women, especially those at risk for breast cancer, should take probiotics to increase the proportion of beneficial bacteria in the breast.”
In addition to prevention, this finding could have potential for helping with the management of patient disease. “It may be possible to increase the abundance of beneficial bacteria at the expense of harmful ones through the use of probiotics,” said Dr. Reid. “Antibiotics targeting harmful bacteria may also be another option for improving breast cancer management. Additional research is warranted to further explore the role of the breast tissue microbiome in the development and prevention of breast cancer.”
“In the near future, I hope we can discover which bacteria or combination of bacteria promote cancer development and which ones could help protect against it, and the mechanisms by which they do so,” said Urbaniak. “The next logical step would then be to unravel how we can use this knowledge to protect women from getting breast cancer or how we can manipulate the microbiome to help treat cancer once a woman gets it.”
The study, “The microbiota of breast tissue and its association with breast cancer”, was featured in Applied and Environmental Microbiology, a journal of the American Society for Microbiology. Dr. Gregor Reid is the Director, Canadian Centre for Human Microbiome and Probiotic Research at Lawson Health Research Institute and a Professor of Surgery, and Microbiology and Immunology at Western University. Urbaniak, who recently completed her PhD at Western University, will soon be a postdoctoral fellow at the NASA Jet Propulsion Laboratory (JPL) in Pasadena, California where she will perform microbiome analyses on samples collected from astronauts as well as those collected from the surfaces of the International Space Station.
Biotech Support Group & Lawson Health Research Institute Enter Collaborative Research Agreement to Monitor Protease Inhibitor Function During COVID-19 Infections
MONMOUTH JUNCTION, NJ USA and LONDON, ON, CANADA – Biotech Support Group and Lawson Health Research Institute announce a collaborative research agreement. The agreement supports further research to monitor protease inhibitor function acquired from blood collected from hospitalized patients with the SARS-CoV-2 (COVID-19) virus. The initial study will be reported as a poster at the upcoming American Society for Mass Spectrometry (ASMS) Conference, Oct. 31 – Nov. 4, 2021, in Philadelphia PA USA. The poster is entitled: “A Preliminary Investigation Using Targeted LC-MS Proteomic Methods Demonstrates Unique Serum Profiles of Hospitalized SARS-CoV-2 Patients. ”
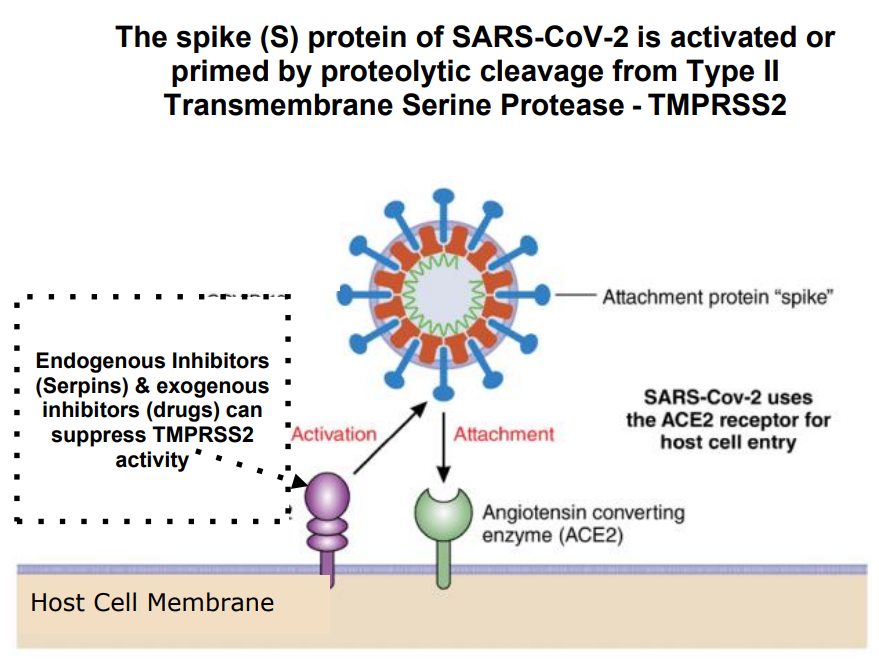
We describe how serine proteases are enzymes that irreversibly split and sever proteins, and thus play critical roles in many key biological processes including digestion, blood coagulation, and immunity. During stress or inflammation, many proteases are produced and discharged into the general circulation, while others are cell membrane-bound; the largest group being the Type II Transmembrane Serine Proteases (TTSPs). TTSPs play a critical cooperative role in the viral infectivity of coronaviruses, especially so now in the SARS-CoV-2 (COVID-19) pandemic.
With such viral infections, there comes an increased level of serine protease activity, due in part to both, an exuberant innate immune response to counter the infectious insult, and the added TTSP activity that derives from the virus load and propagation. As a result, current clinical practice cannot predict and monitor the protease overload triggered by SARS-CoV-2 (COVID-19) and similar infections, and with that, any possible severity, from thrombosis and other complications.
The purpose of this research agreement is to bring together complementary technologies, methods, and intellectual property towards the goals of:
-
utilizing one or more proteomic patterns from Serpin subforms as biomarkers for precision or personalized medicine to clinically manage viral infections; and,
-
developing new therapeutic strategies which can modulate serine proteases, for better clinical outcomes of hospitalized Covid-19 patients.
“This is a very exciting collaboration for us as it supports our efforts in sample prep on the product side, along with bolstering our biomarker intellectual property. This is because, for serum/plasma samples, small biological variances remain hard to measure robustly, mainly due to the presence of highly abundant proteins. For validating small differences established from discovery proteomics, it becomes critical to pair target peptides to sample prep methods to best quantify and establish differentiated profiles between disease and normal states. For this purpose, we will report on the beneficial use of AlbuSorb™ PLUS, to deplete Albumin & IgG in our upcoming ASMS poster, ” states Swapan Roy, Ph.D., President and Founder of Biotech Support Group.
“Based on our preliminary Serpin data profiles, we established differences between the three patient cohorts. This suggests the importance of considering Serpin functions as major players in both risk factors for infection, as well as progressiveness to severe disease. Now through this collaboration, we will investigate larger cohorts using the methods to measure functional Serpin features by LC-MS. Our joint goal is to have biomarkers to help personalize clinical decisions, and develop new anti-COVID-19 therapeutic strategies.” states Douglas Fraser, MD, Ph.D., Lawson Clinician Scientist.
“This very timely collaboration will help to further develop critically needed COVID-19 therapeutic approaches that are included as part of an extensive intellectual property portfolio related to Dr. Fraser’s work on COVID-19 diagnostics, outcome prediction and therapeutic approaches that we have filed over the past year,” states Kirk Brown, Lawson Manager, Business Development at WORLDiscoveries.
Click here to learn more about how BSG’s Sample Prep Products have been used and cited for Discovery, Targeted and Clinical Proteomic applications.
-30-
See all Lawson Media Releases
Converging with cultural and technological disruptions forthcoming in healthcare, Biotech Support Group develops methods for cost effective and efficient sample prep essential for these expanding markets. Following a tiered business strategy, the company continues its growth in the consumable research products area supporting the rapidly expanding installation of LC-MS instrument and computational infrastructure. For this market, key products include: AlbuVoid™ and AlbuSorb ™ for albumin depletion, Cleanascite ™ for lipid adsorption, HemogloBind™ and HemoVoid™ for hemoglobin removal, and NuGel™ for functional proteomics. From these innovations, the company has acquired knowledgebase and biomarker intellectual property assets that support discoveries of protein markers from blood, with special emphasis on personalized medical decisions for cancer and Covid-19 patients. For more information, go to https://www.biotechsupportgroup.com.
Lawson Health Research Institute is one of Canada’s top hospital-based research institutes, tackling the most pressing challenges in health care. As the research institute of London Health Sciences Centre and St. Joseph’s Health Care London, our innovation happens where care is delivered. Lawson research teams are at the leading-edge of science with the goal of improving health and the delivery of care for patients. Working in partnership with Western University, our researchers are encouraged to pursue their curiosity, collaborate often and share their discoveries widely. Research conducted through Lawson makes a difference in the lives of patients, families and communities around the world. To learn more, visit www.lawsonresearch.ca.
WORLDiscoveries is the business development arm of London’s extensive research network and the bridge between local invention and global industry. Born out of a partnership between Western University, Robarts Research Institute and Lawson Health Research Institute (collectively known as the Partners), WORLDiscoveries draws upon a mix of industry connections, sector-specific market knowledge and business development expertise, to help researchers and local inventors commercialize their discoveries through licensing and new company spin-offs.
Senior Media Relations Consultant
Communications & Public Engagement
T: 519-685-8500 ext. 73502
Celine.zadorsky@lhsc.on.ca
Book Launch: From Therapeutic Relationships to Transitional Care
You are invited to a virtual event to learn more about a new publication from Routledge: From Therapeutic Relationships to Transitional Care: A Theoretical and Practical Roadmap. Hear from a panel of speakers who offer different perspectives on this important work.
This publication provides a foundational understanding of therapeutic relationships and the transitional discharge model (TDM) developed in Ontario by a team led by Dr. Cheryl Forchuk, Assistant Scientific Director at Lawson Health Research Institute. This approach is considered a “good practice” by the World Health Organization (WHO) and was recently included in its Guidance on Community Mental Health Services.
The transition from hospital to community is complex and can be challenging for people who have been diagnosed with a mental illness. TDM is a person-centered, evidenced-based approach that creates a safety net by providing seamless support during this transition. Peer support workers play an important role in providing support and links to community-based services based on people’s expressed needs for support.
More about the Transitional Discharge Method, shown in Ontario to be a beneficial and cost-effective approach to care as people are discharged from hospital, preventing re-admission:
Event Details
Date: Wednesday, June 23, 2021
Time: 10 – 11 am
Format: Zoom Webinar and Facebook Live. (You will receive the Zoom link after you’ve registered for the virtual event)
Purchase your copy of the book HERE and enter promo code SMAO3 at checkout for a 20% discount.

Bridging the gap between high school and university
Exposure to professional experiences can help students envision themselves in specific professions and Dr. Donna Goldhawk aims to do just this by introducing medical research to high school science students. As coordinator of Youth Outreach for the Ontario Research Fund (ORF) grant entitled “Heart Failure: Prevention through Early Detection Using New Imaging Methods,” she is pleased to announce the call for applications to the 2019 Summer Studentship program.
This year, a total of three ORF summer scholarships are available at Lawson Health Research Institute (London), University of Ottawa Heart Institute (Ottawa), and Sunnybrook Research Institute (Toronto). Interested applicants can forward their cover letter and resume to @email by May 15, 2019.
Dr. Goldhawk is a Scientist at Lawson Health Research Institute and an Assistant Professor in Medical Biophysics at Western University. Since 2009, she has helped inspire students to pursue research by training them in her lab. In conjunction with secondary schools in the Thames Valley region and the Partners in Experiential Learning (PEL) program, she teaches youth that choosing a scientific career allows for multiple opportunities to fuse interests with a specific area of focus.
The PEL program was introduced in 2004 by Dr. Jim Koropatnick, Lawson Scientist, and Mr. Rodger Dusky, a retired secondary school teacher. PEL has been a long-standing partnership between the Thames Valley District School Board, London District Catholic School Board, Ministry of Education, Western University, London Health Sciences Centre and St. Joseph’s Health Care London.
This unique academic co-op education program identifies high school students interested in research and prepares them for placement in a health research environment. The students earn high school credits and receive training in basic laboratory research. In Dr. Goldhawk’s experience, PEL attracts an excellent caliber of student who is eager to learn, aspiring to pursue further education and capable of contributing to the science of molecular imaging.
Through the ORF, the Ministry of Research and Innovation emphasizes the value of attracting youth to the scientific enterprise. Students can expect experiential learning tailored to their current understanding of science and aimed at expanding this knowledge for a greater appreciation of biomedical research. For example, in Dr. Goldhawk’s laboratory cardiac research is focused on the measurement of iron contrast using MRI.
“Many cells have naturally distinct iron metabolism; however, we are also using genes from magnetic bacteria to improve the magnetic sensitivity of mammalian cells. MRI can then be used to detect disease processes,” states Dr. Goldhawk. “We are particularly interested in reporter gene expression for MRI, a term coined to describe how changes in the pattern of gene activity and its related protein activity may be visualized.”
Her current team not only includes graduate and undergraduate students at various stages in their programs but also an international collaboration with Dr. Weiwei Zhang, a Western Visiting Scholar from China. In this setting, high school students step into a work atmosphere where they can interact with individuals at different levels. As well, molecular imaging research works closely with multiple areas so students acquire training in both biology and physics.
“To retain the interest of talented young scientists, it is essential to provide them with interesting challenges that reward their curiosity and ambition to learn,” explains Dr. Goldhawk. “In hospital-based research institutes, youth are also exposed to the clinical side for which our research is targeted. This is a quality scientific experience for youth interested in expanding their outlook, performance, technical, problem-solving and record-keeping skills. I think it is essential to build these opportunities into our scientific programs.”
Bridging the gap from discovery to patient care
The following column was provided to Hospital News by Dr. David Hill, scientific director, Lawson Health Research Institute.
Dedicated health researchers across the country are working every day to make discoveries that improve patient outcomes and support a higher quality of life. Our research hospitals have a mandate to develop and test new treatments, technologies and procedures that address our most pressing health challenges.
There is a cycle of health innovation that is supposed to happen – scientific discoveries are made, they transition to clinical trials and then are adopted as an improved standard of care. This is followed by evaluations of the new method and moving along to the next cycle of refinements and improvements. But the reality in Canada is that gaps, in some cases chasms, disrupt the creation and adoption of evidence-based health innovations.
This starts with discovery. The research funding climate in Canada continues to be extremely difficult. Despite the Federal Government outlining substantial new investments in discovery research in Budget 2018 much of that money will not be available to researchers until 2020 and beyond, and the Canadian Institutes of Health Research (CIHR) continues to be limited by a low funding rate of around 14 per cent. Funding for large, definitive clinical trials is especially difficult to support with public funds. Too many good ideas are left unfunded, and in that environment researchers can feel it’s wiser to propose incremental and ‘safe’ science that avoids controversy and the risk of losing support for experienced research teams built up over many years.
Yet researchers are persistent and manage to secure funding from a range of smaller agencies. Unfortunately, many of these do not fund the indirect costs of research necessary to cover administration, infrastructure, equipment maintenance and upgrades, and the investigator’s salaries. Indirect costs are estimated to be approximately 40 per cent on top of direct study costs. In research hospitals these costs generally fall on the institutions to find funding outside of the provincial government budget that covers the costs of hospital care. This is a negative spiral whereby the more successful a hospital becomes in attracting research funds, the bigger the gap in finding the resources to support that research.
When innovative solutions to health care problems are delivered, backed by solid evidence, adoption into our hospital-based care is often far from rapid. Currently, in most provinces, there is no funding mechanism to translate health innovation to the ‘real world’ setting of our front-line care. Yet, such a mechanism is crucial to the translation of science. Clinical trials are carefully designed with strict protocols and criteria for a highly specific population of patients. Innovations that work in a clinical trial do not always work in the real world where patient populations and settings are much more complex.
Following a clinical trial, innovation needs to be tested at the point of care. Without funding to do this, research innovation hits a roadblock. Scientists are left waiting and hoping that one day their provincial government might look to incorporating their innovations into the health system funding schedule. Meanwhile, their work may be used to inform health care improvements in other nations.
An example can be found in medical imaging research. Canadian scientists, including those at Lawson Health Research Institute, are leaders in the development of positron emission tomography (PET) biomarkers to improve diagnosis and understanding of disease using PET scans. PET biomarkers are successfully created and then validated in clinical trials across our nation, but there is little funding to translate them back in a timely fashion to patient care in our hospitals. While countries like the US and Europe readily adopt these innovations, Canada lags behind.
This gap was addressed in a report to the Federal government by the Advisory Panel on Healthcare Innovation entitled “Unleashing Innovation: Excellent Healthcare for Canada” and published in 2015. The panel recommended the formation of a Healthcare Innovation Agency of Canada open to hospitals and other care providers in order to evaluate health innovations in the real-world setting of our health system. Scientists would apply by putting forward evidence from their research, including that collected from clinical trials. They would then design a new translational study to test their innovation at the point of care, with the goal of building evidence for presentation to provincial government.
Will everything tested at the point of care succeed? No; some things will fail in the real world. This is why such a fund is so important. It will show whether or not an innovation truly benefits patients and if it’s ready for wider adoption. If successful, the evidence will highlight the right time to bring innovations into the mainstream standard of care. By bridging this gap in the system, we can improve patient care and ensure a timely return on the investment in science.
Dr. David Hill is scientific director at Lawson Health Research Institute, the research institute of London Health Sciences Centre and St. Joseph’s Health Care London.